Nuclear chemistry: Difference between revisions
imported>Mark Rust |
imported>Mark Rust |
||
| Line 23: | Line 23: | ||
Another example is the work which was done on the methylation of elements such as [[sulfur]], [[selenium]], [[tellurium]] and [[polonium]] by living organisms. It has been shown that [[bacteria]] can convert these elements into volitile compounds,<ref>N. Momoshima, Li-X. Song, S. Osaki and Y. Maeda, "Biologically induced Po emission from fresh water", ''Journal of Environmental Radioactivity'', 2002, '''63''', 187-197</ref> it is thought that [[methylcobalamin]] ([[vitamin]] [[B12]] alkylates these elements to create the dimethyls. It has been shown that a combination of [[Cobaloxime]] and inorganic polonium in [[sterile]] water forms a volitile polonium compound, while a control experiment which did not contain the [[cobalt]] compound did not form the volitile polonium compound.<ref>N. Momoshima, Li-X. Song, S. Osaki and Y. Maeda, "Formation and emission of volatile polonium compound by microbial activity and polonium methylation with methylcobalamin", ''Environmetal Science and Technology'', 2001, '''35''', 2956-2960</ref>. For the [[sulfur]] work the isotope <sup>35</sup>S was used, while for polonium <sup>207</sup>Po was used. In some related work the addition of <sup>57</sup>Co to the bacterial culture it was shown that the bacteria convert avaiable cobalt into methylcobalamin. | Another example is the work which was done on the methylation of elements such as [[sulfur]], [[selenium]], [[tellurium]] and [[polonium]] by living organisms. It has been shown that [[bacteria]] can convert these elements into volitile compounds,<ref>N. Momoshima, Li-X. Song, S. Osaki and Y. Maeda, "Biologically induced Po emission from fresh water", ''Journal of Environmental Radioactivity'', 2002, '''63''', 187-197</ref> it is thought that [[methylcobalamin]] ([[vitamin]] [[B12]] alkylates these elements to create the dimethyls. It has been shown that a combination of [[Cobaloxime]] and inorganic polonium in [[sterile]] water forms a volitile polonium compound, while a control experiment which did not contain the [[cobalt]] compound did not form the volitile polonium compound.<ref>N. Momoshima, Li-X. Song, S. Osaki and Y. Maeda, "Formation and emission of volatile polonium compound by microbial activity and polonium methylation with methylcobalamin", ''Environmetal Science and Technology'', 2001, '''35''', 2956-2960</ref>. For the [[sulfur]] work the isotope <sup>35</sup>S was used, while for polonium <sup>207</sup>Po was used. In some related work the addition of <sup>57</sup>Co to the bacterial culture it was shown that the bacteria convert avaiable cobalt into methylcobalamin. | ||
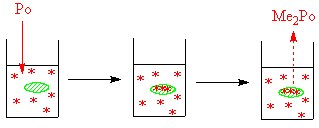
[[Image:Posmissionbygerms.jpg|left|thumb|550px|In the first picture the polonium is added to the culture of the bacteria, by the time that the second picture is shown the bacteria have taken up some of the polonium. In the third picture the bacteria have converted some of the inorganic polonium into the dimethyl form]] | |||
====Environmental==== | ====Environmental==== | ||
Radiochemistry also includes the study of the behaviour of radioisotopes in the environment; for instance, a forest or grass fire can make radioisotopes become mobile again.<ref>Yoschenko VI ''et al'' (2006) Resuspension and redistribution of radionuclides during grassland and forest fires in the Chernobyl exclusion zone: part I. Fire experiments ''J Envir Radioact'' '''86''':143-63 PMID 16213067</ref> In these experiments, fires were started in the exclusion zone around [[Chernobyl]] and the radioactivity in the air downwind was measured. | Radiochemistry also includes the study of the behaviour of radioisotopes in the environment; for instance, a forest or grass fire can make radioisotopes become mobile again.<ref>Yoschenko VI ''et al'' (2006) Resuspension and redistribution of radionuclides during grassland and forest fires in the Chernobyl exclusion zone: part I. Fire experiments ''J Envir Radioact'' '''86''':143-63 PMID 16213067</ref> In these experiments, fires were started in the exclusion zone around [[Chernobyl]] and the radioactivity in the air downwind was measured. | ||
Revision as of 17:59, 12 January 2007
Nuclear chemistry is a subfield of chemistry dealing with radioactivity, nuclear processes and nuclear properties. It includes:
- the chemistry of radioactive elements such as the actinides, radium and radon together with the chemistry associated with equipment (such as nuclear reactors) which are designed to perform nuclear processes. This includes the corrosion of surfaces and the behaviour under conditions of both normal and abnormal operation (such as during an accident). An important area is the behaviour of objects and materials after being placed into a waste store or otherwise disposed of.
- the study of the chemical effects resulting from the absorption of radiation within living animals, plants, and other materials. The radiation chemistry controls much of radiation biology as radiation has an effect on living things at the molecular scale, to explain it another way the raidation alters the biochemicals within an organism, the alteration of the biomolecules then changes the chemistry which occurs within the organism, this change in biochemistry then can lead to a biological outcome. As a result nuclear chemistry greatly assists the understanding of medical treatments (such as cancer radiotherapy) and has enabled these treatments to improve.
- the study of the production and use of radioactive sources for a range of processes. These include radiotherapy in medical applications; the use of radioactive tracers within industry, science and the environment; and the use of radiation to modify materials such as polymers[7][8] .
- the study and use of nuclear processes in non-radioactive areas of human activity. For instance, nuclear magnetic resonance (NMR) spectroscopy is commonly used in synthetic organic chemistry and physical chemistry and for structural analysis in macromolecular chemistry.
Early history
After the discovery of X-rays by Wilhelm Röntgen, many scientists began to work on ionizing radiation. One of these was Henri Becquerel, who investigated the relationship between phosphorescence and the blackening of photographic plates. When Becquerel (working in France) discovered that, with no external source of energy, the uranium generated rays which could blacken (or fog) the photographic plate, radioactivity was discovered. Marie Curie (working in Paris) and her husband Pierre Curie isolated two new radioactive elements from uranium ore. They used radiometric methods to identify which stream the radioactivity was in after each each chemical separation; they separated the uranium ore into each of the different chemical elements that were known at the time, and measured the radioactivity of each fraction. They then attempted to separate these radioactive fractions further, to isolate a smaller fraction with a higher specific activity (radioactivity divided by mass). In this way, they isolated polonium and radium. It was noticed in about 1901 that high doses of radiation could cause an injury in humans, Becquerel had carried a sample of radium in his pocket and as a result he suffered a high localised dose which resulted in a radiation burn[9] this injury resulted in the biological properties of radiation being investigated, which in time resulted in the development of medical treatments. Marie Curie's daughter (Irène Joliot-Curie) and her husband were the first to 'create' radioactivity: they bombarded boron with alpha particles to make a proton-rich isotope of nitrogen; this isotope emitted positrons.[10] In addition, they bombarded aluminium and magnesium with neutrons to make new radioisotopes.
Ernest Rutherford, working in Canada and England, showed that radioactivity decay can be described by a simple equation (a linear first degree derivative equation, now called first order kinetics), implying that a given radioactive substance has a characteristic "half life" (the time taken for the amount of radioactivity present in a source to diminish by half). He also coined the terms alpha, beta and gamma rays, he converted nitrogen into oxygen, and most importantly he supervised the students who did the Geiger-Marsden experiment (gold leaf experiment) which showed that the 'plum pudding model' of the atom was wrong. In the plum pudding model, proposed by J. J. Thomson in 1904, the atom is composed of electrons surrounded by a 'cloud' of positive charge to balance the electrons' negative charge. To Rutherford, the gold foil experiment implied that the positive charge was confined to a very small nucleus leading first to the Rutherford model, and eventually to the Bohr model of the atom, where the positive nucleus is surrounded by the negative electrons.
Main areas
Radiochemistry
Radiochemistry is the chemistry of radioactive materials, where radioactive isotopes of elements are used to study the properties and chemical reactions of non-radioactive isotopes (often within radiochemistry the absence of radioactivity leads to a substance being described as being inactive as the isotopes are stable).
Biochemical uses
One biological application is the study of DNA using radioactive phosphorus-32. In these experiments stable phosphorus is replaced by the chemical identical radioactive P-32, and the resulting radioactivity is used in analysis of the molecules and their behaviour.
Another example is the work which was done on the methylation of elements such as sulfur, selenium, tellurium and polonium by living organisms. It has been shown that bacteria can convert these elements into volitile compounds,[1] it is thought that methylcobalamin (vitamin B12 alkylates these elements to create the dimethyls. It has been shown that a combination of Cobaloxime and inorganic polonium in sterile water forms a volitile polonium compound, while a control experiment which did not contain the cobalt compound did not form the volitile polonium compound.[2]. For the sulfur work the isotope 35S was used, while for polonium 207Po was used. In some related work the addition of 57Co to the bacterial culture it was shown that the bacteria convert avaiable cobalt into methylcobalamin.
Environmental
Radiochemistry also includes the study of the behaviour of radioisotopes in the environment; for instance, a forest or grass fire can make radioisotopes become mobile again.[3] In these experiments, fires were started in the exclusion zone around Chernobyl and the radioactivity in the air downwind was measured.
It is important to note that radioactivity is present everywhere (and has been since the formation of the earth). According to the IAEA, one kilogram of soil typically contains the following amounts of the following three natural radioisotopes 370 Bq 40K (typical range 100-700 Bq), 25 Bq 226Ra (typical range 10-50 Bq), 25 Bq 238U (typical range 10-50 Bq) and 25 Bq 232Th (typical range 7-50 Bq).[4]
Radiation chemistry
Radiation chemistry is the study of the chemical effects of radiation on matter; this is very different to radiochemistry as no radioactivity needs to be present in the material which is being chemically changed by the radiation. An example is the conversion of water into hydrogen gas and hydrogen peroxide.
Reduction of organics by solvated electrons
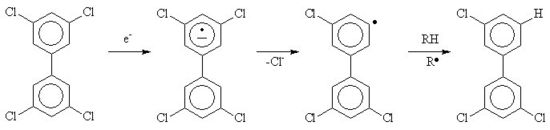
A recent area of work has been the destruction of toxic organic compounds by irradiation [5]; after irradiation, "dioxins" (polychlorodibenzo-p-dioxins) are dechloroinated in the same way as PCBs can be converted to biphenyl an inorganic chloride. This is because the solvated electrons react with the organic compound to form a radical anion, which decomposes by the loss of a chloride anion. If a deoxygenated mixture of PCBs in isopropanol or mineral oil is irradiated with gamma rays, then the PCBs will be dechlorinated to form inorganic chloride and biphenyl. The reaction works best in isopropanol if potassium hydroxide (caustic potash) is added. The base deprotonates the hydroxydimethylmethyl radical to be converted into acetone and a solvated electron, as the result the G value (yield for a given energy due to radiation deposited in the system) of chloride can be increased becuase the radiation now starts a chain reaction, each solvated electron formed by the action of the gamma rays can now convert more than one PCB molecule.[6][7]If oxygen, acetone, nitrous oxide, sulfur hexafluoride or nitrobenzene is present in the mixture, then the reaction rate is reduced. This work has been done recently in the USA, often with used nuclear fuel as the radiation source.[11][12]
In addition to the work on the destruction of aryl chlorides it has been shown that aliphatic chlorine and bromine compounds such as perchloroethylene,[8] Freon (1,1,2-trichloro-1,2,2-trifluoroethane) and halon-2402 (1,2-dibromo-1,1,2,2-tetrafluoroethane) can be dehalogenated by the action of radiation on alkaline isopropanol solutions. Again a chain reaction has been reported.[9]
Reduction of metal compounds
In addition to the reduction of organic compounds by the solvated electrons it has been reported that upon irradation a pertechnetate solution, at pH 4.1 is converted to a colloid of technetium dioxide. Irradation of a solution at pH 1.8 soluble Tc(IV) complexes are formed. Irradation of a solution at 2.7 forms a mixture of the colloid and the soluble Tc(IV) compounds.[10]
It has been shown that the irradation of aqueous solutions of lead compounds leads to the formation of elemental lead, when an inorganic solid such as bentonite and sodium formate are present then the lead is removed from the aqueous solution.[11]
Polymer modifcation
Another key area uses radiation chemistry to modify polymers. Using radiation, it is possible to convert monomers to polymers, to crosslink polymers, and to break polymer chains[13][14]. Both man-made and natural polymers (such as carbohydrates [15]) can be processed in this way.
Water chemistry
Both the harmful effects of radiation upon biological systems (induction of cancer and acute radiation injuries) and the useful effects of radiotherapy involve the radiation chemistry of water. The vast majority of biological molecules are present in an aqueous medium; when water is exposed to radiation, the water absorbs energy, and as a result forms chemically reactive species that can interact with dissolved substances (solutes). Water is ionized to form a solvated electron and H2O+, the H2O+ cation can react with water to form a hydrated proton (H3O+) and a hydroxyl radical (HO.). Furthermore, the solvated electron can recombine with the H2O+ cation to form an excited state of the water, this excited state then decomposes to species such as hydroxyl radicals (HO.), hydrogen atoms (H.) and oxygen atoms (O.). Finally, the solvated electron can react with solutes such as solvated protons or oxygen molecules to form respectively hydrogen atoms and dioxygen radical anions. The fact that oxygen changes the radiation chemistry might be one reason why oxygenated tissues are more sensitive to irradiation than the deoxygenated tissue at the centre of a tumor. The free radicals, such as the hydroxyl radical, chemically modify biomolecules such as DNA, leading to damage such as breaks in the DNA strands. Some substances can protect again radiation-induced damage by reacting with the reactive species generated by the irradiation of the water.
Equipment
Industrial processing equipment
To process materials, either a gamma source or an electron beam can be used. The international type IV (wet storage) irradiator is a common design (the JS6300 and JS6500 gamma sterilizers (made by 'Nordion International'[16], which used to trade as 'Atomic Energy of Canada Ltd') are typical. [12]. In these irradiation plants, the source is stored in a deep well filled with water when not in use. When the source is required, it is moved by a steel wire to the irradiation room where the products which are to be treated are present; these objects are placed inside boxes which are moved through the room by an automatic mechanism. By moving the boxes from one point to another, the contents are given a uniform dose. After treatment, the product is moved by the automatic mechanism out of the room. The irradiation room has very thick concrete walls (about 3m thick) to prevent gamma rays from escaping. The source consists of 60Co rods sealed within two layers of stainless steel, the rods are combined with inert dummy rods to form a rack with a total activity of about 12.6PBq (340kCi).
Research equipment
While it is possible to undertake some types of research using an irradiator much like that used for gamma sterilization, it is common in some areas of science to use a time resolved experiment where a material is subjected to a pulse of radiation (normally electrons from a LINAC. After the pulse of radiation the concentration of different substances within the material are measured by emission spectroscopy or Absorption spectroscopy, hence the rates of reactions can be determined. This allows the relative abilitys of substances to react with the reactive species generated by the action of radiation on the solvent (commonly water) to be measured. This experiment is known as pulse radiolysis[17] which is closely related to Flash photolysis.
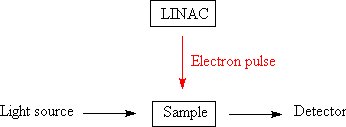
In the latter experiment the sample is excited by a pulse of light before the decay of the excited states is examined by spectroscopy, in this way the excited states of molecules can be examined[[18]] and sometimes the formation of new compounds can be investigated.[13][19] Some of the Flash photolysis work has lead to a greater understanding of the effects of halogen containing compounds upon the ozone layer.[20]
Study of nuclear reactions
see also nuclear physics
A combination of radiochemistry and radiation chemistry is used to study nuclear reactions such as fission and fusion. Some early evidence for nuclear fission was the formation of a shortlived radioisotope of barium which was isolated from neutron irradiated uranium ( 139Ba, with a half-life of 83 minutes and 140Ba, with a half-life of 12.8 days, are major fission products of uranium). At the time, it was thought that this was a new radium isotope, as it was then standard radiochemical practice to use a barium sulphate carrier precipitate to assist in the isolation of radium.[21]. More recently, a combination of radiochemical methods and nuclear physics has been used to try to make new 'superheavy' elements; it is thought that islands of relative stability exist where the nuclides have half-lives of years, thus enabling weighable amounts of the new elements to be isolated. For more details of the original discovery of nuclear fission see the work of Otto Hahn.[14].
Radioisotope production
The processes forming new isotopes (often radioactive) involve several areas of nuclear chemistry.
Processes
- By irradiation with slow neutrons, it is possible to form neutron-rich isotopes which tends to decay by beta decay (i.e. by electron emission from the nuclei). For instance, irradiating 59Co with neutrons forms an excited state of 60Co (best written as 60mCo) which decays by emitting a gamma ray to the ground state of 60Co, and which in turn decays by emitting an electron to form 60mNi. The excited state of the 60mNi then decays with the emission of two gamma photons to the ground state of 60Ni. As the neutron energy increases, the simple capture reactions become less important, while other reactions such as the (n,p) reaction become more important. An example is the production of phosphorus-32 by neutron irradiation of 32S. The sulphur nucleus captures a neutron and emits a proton to form the radioactive phosphorus isotope ( 32P). Carbon-14 is obtained in a similar manner by irradiating 14N with neutrons.
- A beam of fast moving positive particles can be obtained using a cyclotron or a linear accelerator (linac); up to 30MeV protons and deuterons can be obtained this way. The energies of these particles are so high that they can overcome the electrostatic barrier which opposes the entry of positive particles into the nucleus. An example of the use of the (p,n) reaction is the conversion of 103Rh into 103Pd, this can be done by irradiating rhodium foil with protons to form the radioactive palladium isotope. The reaction of beryllium with alpha particles is another example. While the reaction of 9Be with 4He2+ generates 12C, its most important aspect is that it generates neutrons.
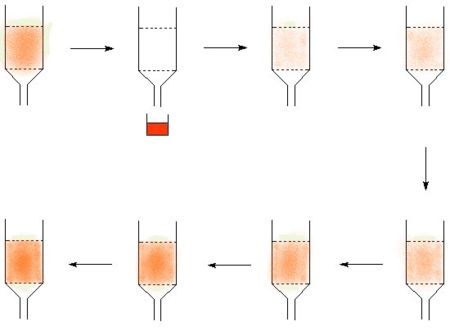
- Many isotopes can be made from a parent isotope which decays to form the desired isotope. If the parent and the product element can be chemically separated, then it is possible to create an "isotope cow". The classic isotope cow is the technetium cow, many others work by the same principle. The technetium cow uses molybdum-99 absorbed on alumina, and it is "milked" by passing saline solution through it to give a solution of technetium.
In the diagram, the technetium is represented in red, in picture two the cow is milked to make a product solution. The technetium then builds up again to allow the cow to return to the technetium loaded state where it can be milked again.
In this way, aqueous solutions of the following isotopes can be made from parent isotopes (shown in brackets)
- 68Ga (68Ge)
- 82Rb (82Sr)
- 99mTc (99Mo)
- 113mIn (113Sn)
- 188Re (188W)
- 62Cu (62Zn)
When the product isotope is a gas, the cow can be milked by allowing the product to diffuse out of a solid. An early way of making radiography sources was to milk radon from a radium source; this method was used by Marie Curie during the first World War (WWI), and was used in the USA to make Brachytherapy sources. By this method, the following isotopes can be obtained from parent isotopes (shown in brackets)
- 81mKr (81Rb)
- 222Rn (226Ra)
In some nuclear materials, new isotopes are formed by the decay of a parent isotope. For instance, the beta decay of 241Pu will form 241Am, so if a sample of plutonium which has been standing for several years is subjected to a new chemical purification, then it is possible to harvest the americium.
- 241Am (241Pu)
Uses
Radioactive sources have many different uses [15]. A sealed source is sealed within a container so that, in normal use, no radioactive material is lost from the source. In many sealed sources, the radioactive filling is surrounded by one or more layers of a corrosion-resistant material (such as stainless steel or gold). Alternatively, it is possible to make a source using material which holds the radioactivity in a chemically resistant and strong form without needing a metal cover. In designing sealed sources, it is common to choose a chemically stable form of the radioactive element, but for cesium radiotherapy sources it is common to use the water soluble chloride, because it is impossible to obtain a high enough density of cesium in any other compound.
- Sealed sources are used for radiotherapy treatment of many cancers as well as for food irradiation, industrial radiography, nuclear gauges and many other applications. In medical radiotherapy, tumors can be treated either by focusing a beam of gamma rays on the area of the body that contains the tumor (teletherapy),, or by surgically placing a smaller radioactive source within or close to the tumor (brachytherapy). The aim is to confine the radiation, as far as possible, to the tumor and to spare healthy tissues in other parts of the body from its effects.
- Open sources are used for a range of applications which include the use of tracers to study the physical operation of industrial processes, to trace the chemical mechanism by which a product forms. For instance, krypton has been used to study the underground combustion of fuels such as oil and coal.[22][23]. They are also used for some forms of radiotherapy. For example, in the treatment of thyroid cancer the patient is given a large dose of 131I. Because the iodine accumulates in the thyroid gland, the tissue of the thyroid gland (and the tummor) suffers a much higher dose of radiation than most of the body. As a result, the radioactive iodine can selectively destroy the thyroid gland and the tumor which is derived from it. Also in terminal care 89Sr is used to destroy bone tumors. [16]
Because cancer cells are more susceptible to being killed by radiation than normal, healthy cells, radiotherapy treatment can be very effective in reducing the bulk of tumors. Radiotherapy is usually accompanied by some form of chemotherapy designed to attack any remaining tumor cells.
Some radiopharmaceuticals are used for medical imaging, including many different technetium complexes [17], while radioactive 201Tl (half-life of 73 hours) is used for diagnostic purposes in nuclear medicine, particularly in stress tests used for risk stratification in patients with coronary artery disease (CAD).[18][19] This isotope of thallium can be generated using a transportable generator which is similar to the technetium cow. The generator contains lead-201 (half life 9.33 hours) which decays by electron capture to the 201Tl. The 201Pb can be produced in a cyclotron by the bombardment of thallium with protons or deuterons by the (p,3n) and (d,4n) reactions.[20]
The nuclear fuel cycle

The chemistry associated with any part of the nuclear fuel cycle, including nuclear reprocessing. The fuel cycle includes all the operations involved in producing fuel, from mining, ore processing and enrichment to fuel production (Front end of the cycle). It also includes the 'in-pile' behaviour (use of the fuel in a reactor) before the back end of the cycle. The back end includes the management of the used nuclear fuel in either a cooling pond or dry storage, before it is disposed of into an underground waste store or reprocessed.
Normal and abnormal conditions
The nuclear chemistry associated with the nuclear fuel cycle can be divided into two main areas, one area is concerned with operation under the intended conditions while the other area is concerned with maloperation conditions where some alteration from the normal operating conditions has occured or (more rarely) an accident is occuring.
The releases of radioactivity from normal operations are the small planned releases from uranium ore processing, enrichment, power reactors, reporcessing plants and waste stores. These can be in a different chemical/physical form to the releases which could occur under accident conditions. In addition the isotope signature of a hypothetical accident may be very different to that of a planned normal operational discharge of radioactivity to the environment.
Release of radioactivity from fuel during normal use and accidents
The IAEA assume that under normal operation the coolant of a water cooled reactor will contain some radioactivity[21] but during a reactor accident the coolant radioactivity level may rise. The IAEA state that under a series of different conditions different amounts of the core inventory can be released from the fuel, the four conditions the IAEA consider are normal operation, a spike in coolant activity due to a sudden shutdown/loss of preasure (core remains covered with water), a cladding failure resulting in the release of the activity in the fuel/cladding gap (this could be due to the fuel being uncovered by the loss of water for 15-30 minutes where the cladding reached a temperture of 650-1250 oC) or a melting of the core (the fuel will have to be uncovered for at least 30 minutes, and the cladding would reach a temperture in excess of 1650 oC).[22]
Based upon the assumption that a PWR contains 300 tons of water, and that the activity of the fuel of a 1 GWe reactor is as the IAEA predict[23], then the coolant activity after an accident such as the three mile island accident where a core is uncovered and then recovered with water then the resulting activity of the coolant can be predicted.
The study of used fuel
Used nuclear fuel is studied in post irradiation examination, where used fuel is examined to know more about the processes that occur in fuel during use, and how these might alter the outcome of an accident. For example, during normal use, the fuel expands due to thermal expansion. This causes cracking, and in extreme cases, such as during the power surge which destroyed the Chernobyl nuclear reactor in April, 1986, the fuel can shatter into very small fragments. Most nuclear fuel is uranium dioxide, which is a cubic solid which has a structure similar to that of calcium fluoride, in used fuel the solid state structure of most of the solid remains the same as that of pure cubic uranium dioxide. SIMFUEL is the name given to the simulated spent fuel which is made by mixing finely ground metal oxides, grinding as a slurry, spray drying it before heating in hydrogen/argon to 1700 oC. [24] In SIMFUEL, 4.1% of the volume of the solid was in the form of metal nanoparticles which are made of molybdenum, ruthenium, rhodium and palladium. Most of these metal particles are of the ε phase (hexagonal) of Mo-Ru-Rh-Pd alloy, while smaller amounts of the α (cubic) and σ (tetragonal) phases of these metals were found in the SIMFUEL. Also present within the SIMFUEL was a cubic perovskite phase which is a barium strontium zirconate (BaxSr1-xZrO3).
Uranium dioxide is very insoluble in water, but after oxidation it can be converted to uranium trioxide or another uranium(VI) compound which is much more soluble. It is important to understand that uranium dioxide (UO2) can be oxidised to an oxygen rich hyperstoichiometric oxide (UO2+x) which can be further oxidised to U4O9, U3O7, U3O8 and UO3.2H2O.
Because used fuel contains alpha emitters (plutonium and the minor actinides), the effect of adding an alpha emitter (238Pu) to uranium dioxide on the leaching rate of the oxide has been investigated. For the crushed oxide, adding 238Pu tended to increase the rate of leaching, but the difference in the leaching rate between between 0.1 and 10% 238Pu was very small. [25]
The concentration of carbonate in the water which is in contact with the used fuel has a considerable effect on the rate of corrosion, because uranium(VI) forms soluble anionic carbonate complexes such as [UO2(CO3)2]2- and [UO2(CO3)3]4-. When carbonate ions are absent, and the water is not strongly acidic, the hexavalent uranium compounds which form on oxidation of uranium dioxide often form insoluble hydrated uranium trioxide phases [26].
By ‘sputtering’, using uranium metal and an argon/oxygen gas mixture, thin films of uranium dioxide can be deposited upon gold surfaces. These gold surfaces modified with uranium dioxide have been used for both cyclic voltammetry and AC impedance experiments, and these offer an insight into the likely leaching behaviour of uranium dioxide. [27]
Fuel cladding interactions
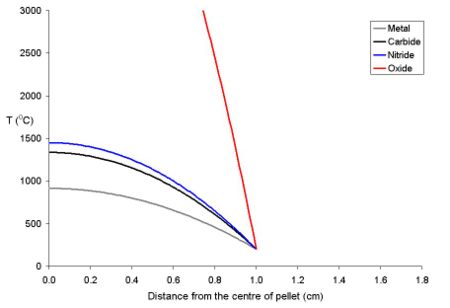
The study of the nuclear fuel cycle includes the study of the behaviour of nuclear materials both under normal conditions and under accident conditions. For example, there has been much work on how uranium dioxide based fuel interacts with the zirconium alloy tubing used to cover it. During use, the fuel swells due to thermal expansion and then starts to react with the surface of the zirconium alloy, forming a new layer which contains both fuel and zirconium (from the cladding). Then, on the fuel side of this mixed layer, there is a layer of fuel which has a higher cesium to uranium ratio than most of the fuel. This is because xenon isotopes are formed as fission products that diffuse out of the lattice of the fuel into voids such as the narrow gap between the fuel and the cladding. After diffusing into these voids, it decays to cesium isotopes. Because of the thermal gradient which exists in the fuel during use, the volatile fission products tend to be driven from the centre of the pellet to the rim area.[28]. Below is a graph of the temperature of uranium metal, uranium nitride and uranium dioxide as a function of distance from the centre of a 20 mm diameter pellet with a rim temperature of 200 oC. It is important to note that the uranium dioxide (because of its poor thermal conductivity) will overheat at the centre of the pellet, while the more thermally conductive other forms of uranium remain below their melting points.
Absorption of fission products on surfaces
Another important area of nuclear chemistry is the study of how fission products interact with surfaces; this is thought to control the rate of release and migration of fission products both from waste containers under normal conditions and from power reactors under accident conditions. It is interesting to note that, like chromate and molybdate, the 99TcO4 anion can react with steel surfaces to form a corrosion resistant layer. In this way, these metaloxo anions act as anodic corrosion inhibitors. The formation of 99TcO2 on steel surfaces is one effect which will retard the release of 99Tc from nuclear waste drums and nuclear equipment which has been lost before decontamination (eg submarine reactors lost at sea). This 99TcO2 layer renders the steel surface passive, inhibiting the anodic corrosion reaction. The radioactive nature of technetium makes this corrosion protection impractical in almost all situations. It has also been shown that 99TcO4 anions react to form a layer on the surface of activated carbon (charcoal) or aluminium.[29][24]. A short review of the biochemical properties of a series of key long lived radioisotopes can be read on line.[25]
It is important to note that 99Tc in nuclear waste may exist in chemical forms other than the 99TcO4 anion, these other forms have different chemical properties.[26]
Similarly, the release of iodine-131 in a serious power reactor accident could be retarded by absorption on metal surfaces within the nuclear plant. [30]
Spinout areas
Some methods first developed within nuclear chemistry and physics have become so widely used within chemistry and other physical sciences that they may be best thought of as separate from normal nuclear chemistry. For example, the isotope effect is used so extensively to investigate chemical mechanisms and the use of cosmogenic isotopes and long-lived unstable isotopes in geology that it is best to consider much of isotopic chemistry as separate from nuclear chemistry.
Kinetics (use within mechanistic chemistry)
The mechanisms of chemical reactions can be investigated by observing how the kinetics of a reaction are changed by making an isotopic modification of a substrate. This is now a standard method in organic chemistry. Briefly, replacing normal hydrogens (protons) by deuteriumwithin a chemical compound causes the rate of molecular vibration (C-H, N-H and O-H bonds show this) to decrease. This then can lead to a decrease in the reaction rate if the rate-determining step involves breaking a bond between hydrogen and another atom. Thus, if the reaction changes in rate when protons are replaced by deuteriums, it is reasonable to assume that the breaking of the bond to hydrogen is part of the step which determines the rate.
Uses within geology, biology and forensic science
Cosmogenic isotopes are formed by the interaction of cosmic rays with the nucleus of an atom. These can be used for dating purposes and for use as natural tracers. In addition, by careful measurement of some ratios of stable isotopes it is possible to obtain new insights into the origin of bullets, ages of ice samples, ages of rocks, and the diet of a person can be identified from a hair or other tissue sample. (See Isotope geochemistry and Isotopic signature for further details).
Biology
Within living things, isotopic labels (both radioactive and nonradioactive) can be used to probe how the complex web of reactions which makes up the metabolism of an organism converts one substance to another. For instance a green plant uses light energy to convert water and carbon dioxide into glucose by photosynthesis. If the oxygen in the water is labeled, then the label appears in the oxygen gas formed by the plant and not in the glucose formed in the chloroplasts within the plant cells.
For biochemical and physiological experiments and medical methods, a number of specific isotopes have important applications.
- Stable isotopes have the advantage of not delivering a radiation dose to the system being studied; however, significant an excess of them in the organ or organisms might still interfere with its functioning, and the availability of sufficient amounts for whole-animal studies is limited for many isotopes. Measurement is also difficult, and ususally requires mass spectroscopy to determine how much of the isotope is present in particular compounds, and there is no means of localizing measurements within the cell.
- H-2 (deuterium), the stable isotope of hydrogen, is a stable tracer, the concentration of which can be measured by mass spectroscopy or NMR. It is incorporated into all cellular structures. Specific deuterated compound can also be produced.
- N-15 the stable isotope of nitrogen, has also been used. It is incorporated mainly into proteins.
- Radioactive isotopes have the advantages of being detectable in very low quantities, in being easily measured by scintillation counting or other radiochemical methods, and in being localizable to particular regions of a cell, and quantifiable by autoradiography. Many compounds with the radioactive atoms in specific positions can be prepared, and are widely available commercially. In high quantities they require precautions to guard the workers from the effects of radiation--and they can easily contaminate laboratory glassware and other equipment. For some isotopes the half-life is so short that preparation and measurement is difficult.
By organic synthesis it is possible to create a complex molecule with a radioactive label that can be confined to a small area of the molecule. For short-lived isotopes such as 11C, very rapid synthetic methods have been developed to permit the rapid addition of the radioactive isotope to the molecule. For instance a palladium catalysed carbonylation reaction in a microfluidic device has been used to rapidly form amides[31] and it might be possible to use this method to form radioactive imaging agents for PET imaging.[27]
- 3H, Tritium, the radioisotope of hydrogen, it available at very high specific activities, and compounds with this isotope in particular positions are easily prepared by standard chemical reactions such as hydrogenation of unsaturated precursors. The isotope emits very soft beta radiation, and can be detected by scintillation counting.
- 11C, Carbon-11 can be made using a cyclotron, boron in the form of boric oxide is reacted with protons in a (p,n) reaction. An alternative route is to react 10B with deuterons. By rapid organic synthesis, the 11C compound formed in the cyclotron is converted into the imaging agent which is then used for PET.
- 14C, Carbon-14 can be made (as above), and it is possible to convert the target material into simple inorganic and organic compounds. In most organic synthesis work it is normal to try to create a product out of two approximately equal sized fragments and to use a convergent route, but when a radioactive label is added, it is normal to try to add the label late in the synthesis in the form of a very small fragment to the molecule to enable the radioactivity to be localised in a single group. Late addition of the label also reduces the number of synthetic stages where radioactive material is used.
- 18F, fluorine-18 can be made by the reaction of neon with deuterons, 20Ne reacts in a (d,4He) reaction. It is normal to use neon gas with a trace of stable flourine (19F2). The 19F2 acts as a carrier which increases the yield of radioactivity from the cyclotron target by reducing the amount of radioactivity lost by absorption on surfaces. However, this reduction in loss is at the cost of the specfic activity of the final product.
Nuclear magnetic resonance (NMR)
NMR spectroscopy uses the net spin of nuclei in a substances upon energy absorption to identify molecules. This has now become a standard spectroscopic tool within synthetic chemistry. One major use of NMR is to determine the bond connectivity within an organic molecule.
NMR imaging also uses the net spin of nuclei (commonly protons) for imaging. This is widely used for diagnostic purposes in medicine, and can provide detailed images of the inside of a person without inflicting any radiation upon them. In a medical setting, NMR is often known simply as "magnetic resonance" imaging, as the word 'nuclear' has negative connotations for many people.
References
- ↑ N. Momoshima, Li-X. Song, S. Osaki and Y. Maeda, "Biologically induced Po emission from fresh water", Journal of Environmental Radioactivity, 2002, 63, 187-197
- ↑ N. Momoshima, Li-X. Song, S. Osaki and Y. Maeda, "Formation and emission of volatile polonium compound by microbial activity and polonium methylation with methylcobalamin", Environmetal Science and Technology, 2001, 35, 2956-2960
- ↑ Yoschenko VI et al (2006) Resuspension and redistribution of radionuclides during grassland and forest fires in the Chernobyl exclusion zone: part I. Fire experiments J Envir Radioact 86:143-63 PMID 16213067
- ↑ Generic Procedures for Assessment and Response during a Radiological Emergency, IAEA TECDOC Series number 1162, published in 2000 [1]
- ↑ Zhao C et al (2007) Radiation Physics and Chemistry, 76:37-45
- ↑ Ajit Singh and Walter Kremers, Radiation Physics and Chemistry, 2002, 65(4-5), 467-472
- ↑ Bruce J. Mincher, Richard R. Brey, René G. Rodriguez, Scott Pristupa and Aaron Ruhter, Radiation Physics and Chemistry, 2002, 65(4-5), 461-465
- ↑ V. Múka, *, R. Silber, M. Pospíil, V. Kliský and B. Bartoníek, Radiation Physics and Chemistry, 1999, 55(1), 93-97
- ↑ Seiko Nakagawa and Toshinari Shimokawa, Radiation Physics and Chemistry, 2002, 63(2), 151-156
- ↑ T. Sekine, H. Narushima, T. Suzuki, T. Takayama, H. Kudo, M. Lin and Y. Katsumura, Colloids and Surfaces A: Physicochemical and Engineering Aspects, 2004, 249(1-3), 105-109
- ↑ M. Pospίšil, V. Čuba, V. Múčka and B. Drtinová, "Radiation removal of lead from aqueous solutions- effects of various sorbants and nitrous oxide", Radiation Physics and Chemistry, 2006, 75, 403-407
- ↑ Features of the design are discussed in the IAEA report on a human error accident in such an irradiation plant [2]
- ↑ George Porter, Nobel lectuure, 11 December 1967
- ↑ Meitner L, Frisch OR (1939) Disintegration of uranium by neutrons: a new type of nuclear reaction Nature 143:239-240 [3]
- ↑ A short review of the use of radioactivity in industry
- ↑ Volkert WA, Hoffman TJ (1999) Therapeutic radiopharmaceuticals Chem Rev 99:2269-92 PMID 11749482
- ↑ Jurisson SS, Lydon JD (1999) Potential technetium small molecule radiopharmaceuticals Chem Rev 99:2205-18 PMID 11749479
- ↑ Thallium Test from Walter Reed Army Medical Center
- ↑ Thallium Stress Test from the American Heart Association
- ↑ Thallium-201 production from Harvard Medical School's Joint Program in Nuclear Medicine
- ↑ page 169 Generic Assessment Procedures for Determining Protective Actions During a Reactor Accident, IAEA-TECDOC-955, 1997
- ↑ page 173 Generic Assessment Procedures for Determining Protective Actions During a Reactor Accident, IAEA-TECDOC-955, 1997
- ↑ page 171 Generic Assessment Procedures for Determining Protective Actions During a Reactor Accident, IAEA-TECDOC-955, 1997
- ↑ A good report on the microstructure of used fuel is Lucuta PG et al (1991) J Nuclear Materials 178:48-60
- ↑ V.V. Rondinella VV et al (2000) Radiochimica Acta 88:527-531
- ↑ For a review of the corrosion of uranium dioxide in a waste store which explains much of the chemistry, see Shoesmith DW (2000) J Nuclear Materials 282:1-31
- ↑ Miserque F et al (2001) J Nuclear Materials 298:280-90
- ↑ Further reading on fuel cladding interactions: Tanaka K et al (2006) J Nuclear Materials 357:58-68
- ↑ Decontamination of surfaces, George H. Goodalland Barry.E. Gillespie, United States Patent 4839100
- ↑ * Glänneskog H (2004) Interactions of I2 and CH3I with reactive metals under BWR severe-accident conditions Nuclear Engineering and Design 227:323-9
- ↑ Miller PW et al (2006) Chemical Communications 546-548
Text books
- Radiochemistry and Nuclear Chemistry
Comprehensive textbook by Choppin, Liljenenzin and Rydberg. ISBN -0750674636, Butterworth-Heinemann, 2001 [28].
- Radioactivity, Ionizing radiation and Nuclear Energy
Basic textbook for undergraduates by Jiri Hála and James D Navratil. ISBN -807302053-X, Konvoj, Brno 2003 [29]
- The Radiochemical Manual
Overview of the production and uses of both open and sealed sources. Edited by BJ Wilson and written by RJ Bayly, JR Catch, JC Charlton, CC Evans, TT Gorsuch, JC Maynard, LC Myerscough, GR Newbery, H Sheard, CBG Taylor and BJ Wilson. The radiochemical centre (Amersham) was sold via HMSO, 1966 (second edition)