Hydrocracking
Hydrocracking is a catalytic chemical process used in petroleum refineries for converting the high-boiling constituent hydrocarbons in petroleum crude oils to more valuable lower-boiling products such as gasoline, kerosene, jet fuel and diesel oil. The process takes place in a hydrogen-rich atmosphere at elevated temperatures (260 – 425 °C) and pressures (35 – 200 bar).[1][2][3]
Basically, the process cracks the high-boiling, high molecular weight hydrocarbons into lower-boiling, lower molecular weight olefinic and aromatic hydrocarbons and then hydrogenates them. Any sulphur and nitrogen present in the hydrocracking feedstock are, to a large extent, also hydrogenated and form gaseous hydrogen sulphide (H2S) and ammonia (NH3) which are subsequently removed. The result is that the hydrocracking products are essentially free of sulfur and nitrogen impurities and consist mostly of paraffinic hydrocarbons.
Hydrocracking plants are capable of processing a wide variety of feedstocks of different characteristics to produce a broad range of products. They can be designed and operated to maximize the production of a gasoline blending component (called hydrocrackate) or to maximize the production of diesel oil.
History
Hydrocracking was first developed in Germany as early as 1915 to provide liquid fuels derived from their domestic coal deposits. The first plant that might be considered as a commercial hydrocracking unit began operation in Leuna, Germany in 1927. Similar efforts to convert coal to liquid fuels took place in the Great Britain, France and other countries.[4][5]
Between 1925 and 1930, Standard Oil of New Jersey collaborated with I.G. Farbenindustrie of Germany to develop hydrocracking technology capable of converting heavy petroleum oils into fuels. Such processes required pressures of 200 – 300 bar and temperatures of over 375 °C and were very expensive.
In 1939, Imperial Chemical Industries of Great Britain developed a two-stage hydrocracking process. During World War II (1939 – 1945), two-stage hydrocracking processes played an important role in producing aviation gasoline in Germany, Great Britain and the United States of America.
After World War II, hydrocracking technology became less important. The availability of petroleum crude oil from the Middle East removed the motivation to convert coal into liquid fuels. Newly developed fluid catalytic cracking processes were much more economical than hydrocracking for converting high-boiling petroleum oils to fuels.
In the early 1960s, hydrocracking become economical for a number of reasons:
- The automobile industry began manufacturing higher-performing automobiles that required high-octane gasoline.
- Fluid catalytic cracking expanded rapidly to meet the demand for high-octane gasoline. However, fluid catalytic cracking, in addition to producing gasoline, produces a by-product high-boiling oil called cycle oil that is very difficult to recycle for further cracking. However, hydrocracking can crack that cycle oil.
- The switch from railroad steam engines to diesel engines and the introduction of commercial jet aircraft in the 1950's increased the demand for diesel oil and for jet fuel. The flexibility of hydrocracking to produce either gasoline, jet fuel or diesel oil made it desirable for petroleum refineries to install hydrocrackers.
- Zeolite-based catalysts, developed and commercialized during the period from about 1964 to 1966, performed much better than the earlier catalysts. Most importantly, they permitted operation at lower pressures than possible with the earlier catalysts. The higher performance and lower operating pressures made possible by the new catalysts resulted in significantly more economical hydrocrackers.
Hydrocracking enjoyed rapid growth in the United States during the late 1960s and the early 1970s. By the mid-1970s, hydrocracking had become a mature process and its growth began to moderate. From then on, hydrocracking growth in the United States proceeded at a slow pace. However, at the same time, hydrocracking enjoyed significant growth in Europe, the Asia-Pacific region and the Middle East.
As of 2001, there were about 155 hydrocracker units operating worldwide[1] and processing about 4,000,000 barrels (550,000 metric tons) per day of feedstock.[6] As of 2009, The feedstock processing capacity of the hydrocrackers in the United States was 1,740, 000 barrels (238,000 metric tons) per day.[7]
Process configurations and a typical flow diagram
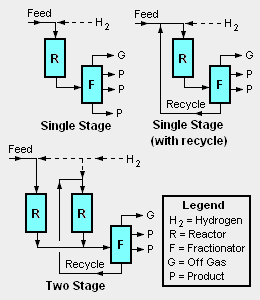
There are a good many different proprietary hydrocracker designs available for use under license as is the case for many of the other processes used in petroleum refineries. There are also a number of different hydrocracker process equipment configurations, the most common of which are depicted in the adjacent diagram:
- Single stage, once through hydrocracker: This configuration uses only one reactor and any uncracked residual hydrocarbon oil from the bottom of the reaction product fractionation (distillation) tower is not recycled for further cracking. For single stage hydrocracking, either the feedstock must first be hydrotreated to remove ammonia and hydrogen sulfide or the catalyst used in the single reactor must be capable of both hydrotreating and hydrocracking.[1]
- Single stage hydrocracker with recycle: This is the most commonly used configuration. The uncracked residual hydrocarbon oil from the bottom of reaction product fractionation tower is recycled back into the single reactor for further cracking. Again, for single stage hydrocracking, either the feedstock must first be hydrotreated to remove ammonia and hydrogen sulfide or the catalyst used in the single reactor must be capable of both hydrotreating and hydrocracking.[1]
- Two stage hydrocracker: This configuration uses two reactors and the residual hydrocarbon oil from the bottom of reaction product fractionation tower is recycled back into the second reactor for further cracking. Since the first stage reactor accomplishes both hydrotreating and hydrocracking, the second stage reactor feed is virtually free of ammonia and hydrogen sulfide. This permits the use of high performance noble metal (palladium, platinum) catalysts which are susceptible to poisoning by sulfur or nitrogen compounds.[1]
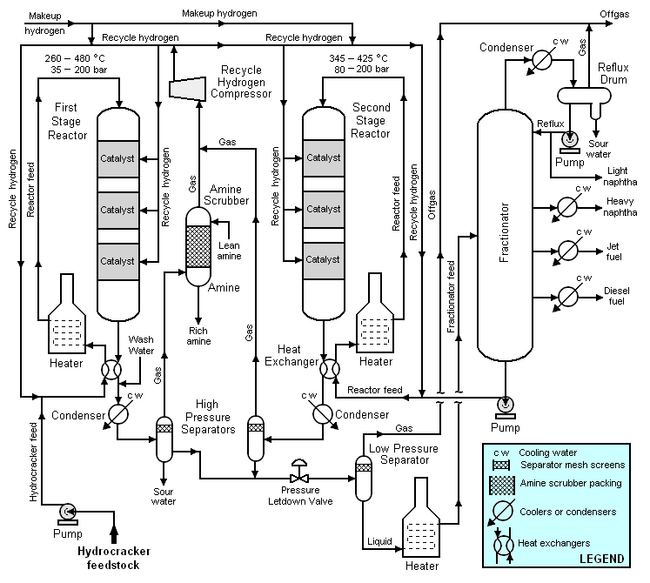
Typical flow diagram of a two stage hydrocracker
The high-boiling, high molecular weight hydrocarbons used as feedstocks for catalytic hydrocrackers include what are commonly referred to as atmospheric gas oil from atmospheric crude oil distillation units, vacuum gas oil from vacuum distillation units, delayed coking gas oil from delayed coking units and cycle oil from fluid catalytic cracking units. For describing the hydrocracking process depicted in the typical flow diagram below, the feedstock will be referred to as simply gas oil.
The gas oil from the feedstock pump is mixed with a stream of high-pressure hydrogen and then flows through a heat exchanger where it is heated by the hot effluent reaction products from the hydrocracker's first stage reactor. The feedstock is then heated further in a fuel-fired heater before it enters the top of first stage reactor and flows downward through three beds of catalyst. The temperature and pressure conditions in the first stage reactor depend upon the specific licensed hydrocracker design, the feedstock properties, the desired products, the catalyst being used and other variables. As a broad generality, the pressure in the first stage reactor may range from 35 to 200 bar and the temperature may range from 260 to 480 °C.
After the effluent reaction product stream from the reactor bottom is cooled by the incoming gas oil feedstock, it is injected with wash water, partially condensed in a water-cooled condenser and routed into a high-pressure vapor-liquid separator for separation into three phases: hydrogen-rich gas, hydrocarbon liquid and water. Sulfur and nitrogen compounds in the gas oil feedstock are converted into gaseous hydrogen sulfide and ammonia by the hydrogenation that takes place in the first stage reactor. The purpose of the wash water is to dissolve some of the hydrogen sulfide and ammonia gases present in the first stage reaction product stream. The resulting aqueous solution of ammonium hydrosulfide (NH4HS) is referred to as sour water and is typically routed to a sour water stripper elsewhere in the petroleum refinery. The sour water stripper removes hydrogen sulfide from the sour water and that hydrogen sulfide is subsequently converted to endproduct elemental sulfur in a Claus process unit.
The hydrogen-rich gas from the high-pressure separator is routed through an amine scrubber where it is contacted with an aqueous amine solution[8] to absorb and remove residual hydrogen sulfide in the gas. The rich amine solution (containing the absorbed hydrogen sulfide) is typically routed to a central amine gas treating unit elsewhere in the refinery.
The hydrocarbon liquid phase from the high-pressure separator flows through a pressure letdown (i.e., pressure reduction) valve and into a low-pressure separator. The reduction in pressure partially vaporizes (see flash evaporation) the liquid. The resulting vapor (referred to as offgas) is routed to a central amine gas treating unit elsewhere in the refinery. The hydrocracked the endproducts of hydrocarbon liquid phase from the low-pressure separator is heated in a fuel-fired heater and fed into the fractionator.
The fractionator is a continuous distillation tower that separates the hydrocracked hydrocarbon stream into naphtha, jet fuel (or kerosene) and diesel oil. The offgas from the tower's associated reflux drum joins the offgas from the low-pressure separator.
Not all of the feedstock hydrocarbons to the first stage reactor are hydrocracked (i.e., converted) into lower-boiling, lower molecular weight hydrocarbons. The bottom stream from the fractionator consists of the unconverted hydrocarbons from the first stage reactor and that stream is mixed with high pressure hydrogen and recycled as feed to the second stage reactor. It is first heated by the hot effluent reaction products from the second stage reactor. The recycled feed is then heated further in a fuel-fired heater before it enters the top of second stage reactor and flows downward through three beds of catalyst. The temperature and pressure conditions in the second stage reactor depend upon the same variables as determine the conditions in the first stage reactor. As a broad generality, the pressure in the second stage reactor may range from 80 to 200 bar and the temperature may range from 345 to 425 °C.
After the effluent reaction product stream from the second stage reactor bottom is cooled by the incoming recycle feed, it is partially condensed in a water-cooled condenser and routed into second high-pressure vapor-liquid separator for separation into two phases: hydrogen-rich gas and hydrocarbon. No water washing of the second stage reactor effluent is needed because the second stage reactor effluent is essentially free of hydrogen sulfide and ammonia gases. For the same reason, the gas from the second high-pressure separator does not require amine scrubbing to remove hydrogen sulfide.
The two hydrogen-rich gas streams (the amine-scrubbed gas from the first high-pressure separator and the gas from second high-pressure separator) are joined and then compressed and recycled for use in both the first and second stage reactor systems.
The hydrogenation of sulfur and nitrogen compounds in the first stage reactor requires the consumption of hydrogen. Likewise, the saturation of olefins and aromatics, in both the first stage and second stage reactors, to form paraffinic hydrocracked products consumes hydrogen. To a large extent, the amount of hydrogen consumption depends on the feedstock content of sulfur, nitrogen, olefins and aromatics. As a broad generality, the consumption of hydrogen in a hydrocracker may range from 1,000 to 3,000 standard cubic feet per barrel of feedstock (195 to 585 normal cubic metres per metric ton of feedstock).[9]
Chemistry and catalysts
Basically, catalytic hydrocracking involves three primary chemical processes:
- Cracking of high-boiling, high molecular weight hydrocarbons found in petroleum crude oil into lower-boiling, lower molecular weight hydrocarbons.
- Hydrogenating unsaturated hydrocarbons (whether present in the original feedstock or formed during the cracking of the high-boiling, high molecular weight feedstock hydrocarbons) to obtain saturated hydrocarbons usually referred to as paraffins or alkanes.
- Hydrogenating any sulfur, nitrogen or oxygen compounds in the original feedstock into gaseous hydrogen sulfide, ammonia and water.
The above primary processes include too many complex reactions to describe each of them in detail. The following four reactions are provided as examples of those complex reactions:[10]
- Reaction 1: Addition of hydrogen to aromatics converts them into hydrogenated rings. These are then readily cracked using acid catalysts.
- Reaction 2: Acid catalyst cracking opens paraffinic rings, breaks larger paraffins into smaller pieces and creates double bonds.
- Reaction 3: Addition of hydrogen to olefinic double bonds to obtain paraffins.
- Reaction 4: Isomerization of branched and straight-chain paraffins.
Hydrocracking catalysts consist of active metals on solid, acidic supports and have a dual function, specifically a cracking function and a hydrogenation function. The cracking function is provided by the acid catalyst support and the hydrogenation function is provided by the metals.[4][11]
The solid acidic support consists of amorphous oxides such as silica-alumina, crystalline zeolite or a mixture of amorphous oxides and crystalline zeolite. Cracking and isomerization reactions (reactions 2 and 4 above) take place on the acidic support. Metals provide the hydrogenation reactions (reactions 1 and 3 above).
The metals that provide the hydrogenation functions can be the noble metals palladium and platinum or the base metals (i.e., non-noble metals) molybdenum, tungsten, cobalt or nickel.
Catalyst cycle life has a major impact on the economics of hydrocracking. Cycles can be as short as 1 year or as long as 5 years. Two years are typical.
References
- ↑ Jump up to: 1.0 1.1 1.2 1.3 1.4 David S.J. Jones and Peter P.Pujado (Editors) (2006). Handbook of Petroleum Processing, First Edition. Springer. ISBN 1-4020-2819-9.
- ↑ James H. Gary and Glenn E. Handwerk (1984). Petroleum Refining: Technology and Economics, 2nd Edition. Marcel Dekker. ISBN 0-8247-7150-8.
- ↑ Editorial Staff (November 2002). "Refining Processes 2002". Hydrocarbon Processing : pages 115 – 117.
- ↑ Jump up to: 4.0 4.1 Julius Scherzer and A.J. Gruia (1996). Hydrocracking Science and Technology, 1st Edition. CRC Press. ISBN 0-8247-9760-4. (This book was the source for most of the History section of this article)
- ↑ Hydrocracking (From the website of Chemical Engineering Resources, which also provided some of this historical information)
- ↑ J.G. Speight and Baki Ozum (2002). Petroleum Refining Processes. Marcel Dekker. ISBN 0-8247-0599-8.
- ↑ Number and Capacity of Petroleum Refineries From the website of the U.S. Energy Information Administration (U.S. EIA), using the drop down Data Series menu to select Catalytic Hydrocracking Charge Capacity.
- ↑ The amines most commonly used for removing hydrogen sulfide from refinery gases area monoethanol amine (MEA), diethanolamine (DEA) and methyldiethanolamine (MDEA)
- ↑ The standard cubic feet of hydrogen are at 60 °F and the normal cubic metres are at 0 °C, both at 1 atmosphere of absolute temperature.
- ↑ John S. Magee and Geoffrey E. Dolbear. Petroleum Catalysts in Nontechnical Language. Pennwell Books. 0-87814-661-X.
- ↑ Jorge Ancheyta and James G. Speight (2007). Hydroprocessing of Heavy Oils and Residua, 1st Edition. CRC Press. ISBN 0-8493-7419-7.