Search results
Jump to navigation
Jump to search
Page title matches
- |elName=Nitrogen '''Nitrogen''' is a [[Chemical elements|chemical element]], typically found as a [[gas]3 KB (539 words) - 18:53, 5 January 2021
- 12 bytes (1 word) - 03:45, 11 November 2007
- #REDIRECT [[Talk:Nitrogen mustards]]36 bytes (4 words) - 15:47, 5 February 2009
- 12 bytes (1 word) - 05:34, 9 November 2007
- 12 bytes (1 word) - 15:14, 27 June 2008
- | pagename = Nitrogen | abc = Nitrogen783 bytes (69 words) - 06:06, 15 March 2024
- OK to delete this picture -- won't be needed for [[Nitrogen cycle]] article. It was a nice try, though, wasn't it?--[[User:Thomas Wrigh192 bytes (31 words) - 21:58, 5 April 2010
- '''Isotopes of Nitrogen and their Physical Properties'''2 KB (267 words) - 13:31, 25 April 2008
- 170 bytes (24 words) - 14:02, 3 March 2010
- == Start [[Nitrogen cycle]] == Start [[Nitrogen cycle]] as stub. [[User:Anthony.Sebastian|Anthony.Sebastian]] 04:33, 21 Feb1 KB (215 words) - 10:32, 26 May 2010
- '''Nitrogen mustard''' is the name of a family of [[alkylating agent]]s, originally dev Additional nitrogen mustards used in medicine include:647 bytes (77 words) - 17:05, 22 June 2009
- {{Image|Nitrogen Cycle drawing.png|right|420px|The nitrogen cycle is a complex system necessary for living beings.}} ...ame=nasa1>[http://soil.gsfc.nasa.gov/NFTG/nitrocyc.htm The Nitrogen Cycle: Nitrogen Transformations in Soil, Water, and Air.] Soil Education Project. National21 KB (3,189 words) - 15:35, 3 September 2010
- 81 bytes (10 words) - 19:22, 21 April 2010
- 4 bytes (0 words) - 23:25, 9 June 2008
- 12 bytes (1 word) - 15:47, 5 February 2009
- #REDIRECT [[Nitrogen cycle]]28 bytes (3 words) - 11:45, 27 March 2010
- 29 bytes (2 words) - 15:49, 5 February 2009
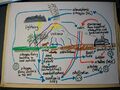
File:Nitrogen cycle.jpg (3,472 × 2,604 (1.65 MB)) - 19:58, 11 March 2022- A complex process in which the element [[nitrogen]] moves into the air, back down into the soil, is changed chemically by var261 bytes (41 words) - 12:23, 27 March 2010
- <includeonly>Non-Metal</includeonly><noinclude>Nitrogen is a [[Non-Metal]].</noinclude>87 bytes (9 words) - 05:50, 6 March 2024
- #REDIRECT [[Nitrogen/Periodic table of elements]]49 bytes (6 words) - 08:26, 6 March 2024
- ...ylating agent]]s derived from [[mustard gas]], with the sulfur replaced by nitrogen, originally [[chemical weapon]]s but now used as highly toxic [[antineopla251 bytes (34 words) - 15:48, 5 February 2009
- 11 bytes (1 word) - 13:06, 17 April 2011
- | pagename = Nitrogen mustards | abc = Nitrogen mustards2 KB (228 words) - 17:05, 22 June 2009
- Auto-populated based on [[Special:WhatLinksHere/Nitrogen]]. Needs checking by a human.3 KB (457 words) - 12:49, 15 March 2024
- 805 bytes (105 words) - 06:57, 6 March 2024
- 34 bytes (7 words) - 18:04, 27 July 2008
- | pagename = Nitrogen cycle | abc = Nitrogen cycle2 KB (291 words) - 10:53, 26 May 2010
- 264 bytes (35 words) - 16:43, 17 May 2010
- ...ctive [[oxidizer]] of colorless dinitrogen tetraoxide (N2O4) and red-brown nitrogen dioxide (NO2) in a temperature-dependent equilibrium; used as a [[hypergoli340 bytes (46 words) - 19:22, 21 April 2010
- 1 bytes (0 words) - 23:24, 9 June 2008
- | pagename = Nitrogen mustard | abc = Nitrogen mustard2 KB (231 words) - 04:31, 28 April 2009
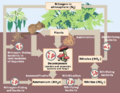
File:Nitrogen Cycle drawing.png (475 × 370 (127 KB)) - 19:56, 11 March 2022- #REDIRECT [[Nitrogen cycle]]28 bytes (3 words) - 11:45, 27 March 2010
- 10 bytes (1 word) - 13:05, 17 April 2011

File:Terra Nitrogen Plant.jpg (364 × 213 (26 KB)) - 19:57, 11 March 2022- 3 bytes (1 word) - 13:00, 10 June 2008
- #REDIRECT [[Nitrogen cycle]]28 bytes (3 words) - 11:46, 27 March 2010
- 1 bytes (1 word) - 17:59, 10 June 2008
- *{{CZ:Ref:Fields 2004 Global nitrogen: cycling out of control}}76 bytes (10 words) - 09:01, 25 August 2010
- {{r|Nitrogen}}176 bytes (21 words) - 04:57, 18 April 2010
- 71 bytes (8 words) - 00:34, 19 April 2010
- Auto-populated based on [[Special:WhatLinksHere/Nitrogen mustards]]. Needs checking by a human.525 bytes (65 words) - 19:02, 11 January 2010
- 43 bytes (8 words) - 05:02, 6 June 2009
- *[http://soil.gsfc.nasa.gov/NFTG/nitrocyc.htm Nitrogen Cycle: Nitrogen Transformations in Soil, Water, and Air] From a page in the [[National Aero ...://www.britannica.com/EBchecked/topic-art/416271/118424/The-nitrogen-cycle Nitrogen cycle] From a page in the Britannica Encyclopedia.352 bytes (46 words) - 05:14, 18 April 2010
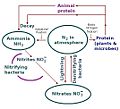
File:Nitrogen Cycle by T Sulcer.jpg (535 × 487 (37 KB)) - 19:54, 11 March 2022- 44 bytes (6 words) - 21:59, 5 April 2010
- 34 bytes (4 words) - 09:00, 25 August 2010
Page text matches
- *[http://soil.gsfc.nasa.gov/NFTG/nitrocyc.htm Nitrogen Cycle: Nitrogen Transformations in Soil, Water, and Air] From a page in the [[National Aero ...://www.britannica.com/EBchecked/topic-art/416271/118424/The-nitrogen-cycle Nitrogen cycle] From a page in the Britannica Encyclopedia.352 bytes (46 words) - 05:14, 18 April 2010
- ...for the mono-nitrogen oxides NO and NO<sub>2</sub> ([[nitric oxide]] and [[nitrogen dioxide]])158 bytes (22 words) - 14:06, 28 May 2011
- |[[Nitrogen]]: | [[Nitrogen/Boiling point|{{:Nitrogen/Boiling point}}]]1 KB (141 words) - 12:54, 26 April 2009
- |[[Nitrogen]]: | {{:Nitrogen/Boiling point}}1 KB (158 words) - 18:09, 23 June 2008
- ...and '''5''', are also less stable because they contain only three nitrogen-nitrogen bonds.1 KB (161 words) - 03:43, 22 November 2023
- *[http://soil.gsfc.nasa.gov/NFTG/nitrocyc.htm#nitrogen%20cycle The Nitrogen Cycle] From a website page of the [[National Aeronautics and Space Administ *[http://www.visionlearning.com/library/module_viewer.php?mid=98 The Nitrogen Cycle] John Arthur Harrison. From the website of Visionlearning, an educat620 bytes (87 words) - 22:32, 11 March 2010
- #REDIRECT [[Nitrogen cycle]]28 bytes (3 words) - 11:45, 27 March 2010
- #REDIRECT [[Nitrogen cycle]]28 bytes (3 words) - 11:45, 27 March 2010
- #REDIRECT [[Nitrogen cycle]]28 bytes (3 words) - 11:46, 27 March 2010
- #REDIRECT [[Talk:Nitrogen mustards]]36 bytes (4 words) - 15:47, 5 February 2009
- #REDIRECT [[Nitrogen/Periodic table of elements]]49 bytes (6 words) - 08:26, 6 March 2024
- (1749 - 1815) Scottish chemist, best known for the discovery of nitrogen.109 bytes (13 words) - 09:53, 27 January 2009
- An organic compound containing a carbon triple bonded to nitrogen.102 bytes (13 words) - 20:19, 29 November 2009
- *{{CZ:Ref:Fields 2004 Global nitrogen: cycling out of control}}76 bytes (10 words) - 09:01, 25 August 2010
- A cyclized chemical with [[nitrogen]], [[oxygen]] or [[sulphur]] within the ring structure.127 bytes (15 words) - 09:16, 6 March 2024
- five-membered rings with two heteroatoms, at least one of which is a nitrogen atom.119 bytes (17 words) - 12:53, 17 May 2008
- | pagename = Nitrogen | abc = Nitrogen783 bytes (69 words) - 06:06, 15 March 2024
- '''Nitrogen mustard''' is the name of a family of [[alkylating agent]]s, originally dev Additional nitrogen mustards used in medicine include:647 bytes (77 words) - 17:05, 22 June 2009
- *[[Nitrogen]]215 bytes (17 words) - 09:15, 6 March 2024
- {{r|Nitrogen}} {{r|Nitrogen cycle}}688 bytes (86 words) - 22:43, 11 March 2010
- Any chemical compound with an [[alkene]] group (C=C) next to a [[nitrogen]] atom ([[amine]] form).134 bytes (19 words) - 20:55, 18 March 2011
- ...ed blood chemistry tests: electrolytes, glucose, creatinine and blood urea nitrogen142 bytes (18 words) - 00:13, 17 February 2009
- Organ system that passes nutrients, gases, hormones, blood cells, nitrogen waste products, etc. to and from cells in the body.163 bytes (23 words) - 06:02, 7 September 2009
- An organic chemical containing a nitrogen atom with tetrahedral arrangement of it electron pairs and its lone pair of164 bytes (23 words) - 18:51, 14 June 2008
- ...aining a [[nitrogen]] atom ("aza"), and [[oxazole]]s, which contain both a nitrogen and an [[oxygen]] atom in the ring system. The [[ketone]] carbon is noted b786 bytes (108 words) - 14:35, 26 November 2010
- ...t composed of equal parts of [[hydrazine]] and UDMH; [[hypergolic]] with [[nitrogen tetroxide]] or [[RFNA]] oxidizer186 bytes (23 words) - 04:42, 21 March 2024
- A [[chemical compound]] (NH<sub>3</sub>) of [[nitrogen]] and [[hydrogen]], occurring as a [[gas]] with a characteristic [[odour]]196 bytes (24 words) - 09:18, 18 March 2010
- ...nary ammonium cation consisting of four ethyl groups attached to a central nitrogen atom, which acts as a potassium-selective ion channel blocker.189 bytes (25 words) - 10:30, 6 September 2009
- === Nitrogen metabolism === {{r|Nitrogen cycle}}:2 KB (258 words) - 22:05, 25 September 2008
- '''Ammonia''' is a [[chemical compound]] (NH<sub>3</sub>) of [[nitrogen]] and [[hydrogen]], occurring as a [[gas]] with a characteristic [[odour]]190 bytes (24 words) - 02:00, 13 January 2024
- ...-lived [[positron]]-emitting [[isotope|radionuclides]] (such as carbon-11, nitrogen-13, oxygen-15 and fluorine-18) to measure cell metabolism.<noinclude>{{DefM279 bytes (30 words) - 02:19, 15 May 2010
- == Start [[Nitrogen cycle]] == Start [[Nitrogen cycle]] as stub. [[User:Anthony.Sebastian|Anthony.Sebastian]] 04:33, 21 Feb1 KB (215 words) - 10:32, 26 May 2010
- An [[antineoplastic agent]], of the [[nitrogen mustard]] family of [[alkylating agent]]s, used as a second-line agent agai204 bytes (27 words) - 15:37, 5 February 2009
- |elName=Nitrogen '''Nitrogen''' is a [[Chemical elements|chemical element]], typically found as a [[gas]3 KB (539 words) - 18:53, 5 January 2021
- <includeonly>Non-Metal</includeonly><noinclude>Nitrogen is a [[Non-Metal]].</noinclude>87 bytes (9 words) - 05:50, 6 March 2024
- ...ylating agent]]s derived from [[mustard gas]], with the sulfur replaced by nitrogen, originally [[chemical weapon]]s but now used as highly toxic [[antineopla251 bytes (34 words) - 15:48, 5 February 2009
- {{cite journal| author=Fields S| title=Global nitrogen: cycling out of control | journal=Environ Health Perspect | year= 2004 | vo268 bytes (35 words) - 08:59, 25 August 2010
- ...hylamine]], CH<sub>3</sub>NH<sub>2</sub>, is the smallest amine. When the nitrogen atom is bonded to one, two, three or four carbon atoms, the chemical is cal1 KB (198 words) - 22:22, 2 December 2008
- ...ivatives. There are also ionic amides, which are very strong bases and the nitrogen equivalents of [[hydroxide]], with the general formula R<sub>2</sub>N<sup>- ...the nitrogen. It also lends partial double bond character to the carbonyl-nitrogen bond, limiting rotation around it. These reactivity patterns are true of am3 KB (410 words) - 02:51, 17 October 2013
- ...rrel Medic''' and used as an annual forage legume. It forms symbioses with nitrogen-fixing rhizobia and mycorrhizal fungi.218 bytes (30 words) - 08:51, 22 September 2008
- A complex process in which the element [[nitrogen]] moves into the air, back down into the soil, is changed chemically by var261 bytes (41 words) - 12:23, 27 March 2010
- ...ent parts of the infrared band; operated as a thermoelectrically or liquid nitrogen cooled device260 bytes (36 words) - 23:57, 12 March 2011
- ...of [[organic chemistry]] — from the [[chemical element|elements]] [[nitrogen]] and [[hydrogen]].258 bytes (31 words) - 08:37, 4 March 2010
- ...Nitrogen Plant.jpg/credit|{{Terra Nitrogen Plant.jpg/credit}}<br/>|}}Terra Nitrogen Plant in Verdigris, Oklahoma that produces ammonia and ammonia derived fert2 KB (263 words) - 08:42, 4 June 2010
- ...ide]]s and [[mercury]]. Typically, more than two-thirds of the flue gas is nitrogen. ...a or urea. In either case, the aim is to produce nitrogen gas, rather than nitrogen oxides.2 KB (391 words) - 15:45, 8 November 2011
- This new article could perhaps use some expansion and the section on the "Nitrogen cycle" could use a review by a biology editor to see that it is worded corr == Revisions of expansion of "Nitrogen cycle" section. ==3 KB (554 words) - 21:00, 27 March 2010
- An example is the reaction of [[nitrogen monoxide]] with [[ozone]] giving [[nitrogen dioxide]] (NO<sub>2</sub>) (in an excited state) and dioxygen (O<sub>2</sub ...ctronic with vibrational motions) the electromagnetic radiation emitted by nitrogen dioxide has a range of wavelengths; however, the emission is centered aroun2 KB (240 words) - 10:54, 7 May 2010
- OK to delete this picture -- won't be needed for [[Nitrogen cycle]] article. It was a nice try, though, wasn't it?--[[User:Thomas Wrigh192 bytes (31 words) - 21:58, 5 April 2010
- ...r [[Hodgkin's disease]] and [[non-Hodgkin's lymphoma]], of the family of [[nitrogen mustards]] in the class of [[alkylating agents]].217 bytes (26 words) - 15:59, 5 February 2009
- ...emical bond]] involving a [[hydrogen]] [[atom]] and either [[Fluorine]], [[Nitrogen]], or [[Oxygen]].203 bytes (25 words) - 23:44, 16 July 2008
- ...ctive [[oxidizer]] of colorless dinitrogen tetraoxide (N2O4) and red-brown nitrogen dioxide (NO2) in a temperature-dependent equilibrium; used as a [[hypergoli340 bytes (46 words) - 19:22, 21 April 2010
- ...orless, odorless and tasteless mixture of [[gas]]es consisting mostly of [[nitrogen]] (N<sub>2</sub>) and [[oxygen]] (O<sub>2</sub>) which is the part of [[Ear326 bytes (50 words) - 14:26, 11 March 2010
- ...s of elements that form diatomic molecules in the gas phase are oxygen and nitrogen (the main components of air), as well as hydrogen and at least four of the332 bytes (53 words) - 21:15, 10 November 2020
- *[[Blood urea nitrogen]]430 bytes (58 words) - 00:17, 17 February 2009
- ...ium (mmol/L)} + \frac{\text{glucose (mg/dl)}}{18} + \frac{\text{blood urea nitrogen (mg/dl)}}{2.8}</math>263 bytes (37 words) - 21:10, 9 September 2020
- * 0.3 (pyridyl nitrogen) * 10.3 for the pyrrole nitrogen1 KB (212 words) - 07:10, 9 June 2009
- {{r|Nitrogen}}196 bytes (24 words) - 22:21, 6 August 2008
- ...en obtained by chemical means, he isolated argon in 1895 from atmospheric nitrogen, thus explaining its higher density by the presence of small amounts of arg2 KB (306 words) - 15:17, 18 August 2009
- {{r|Nitrogen}}176 bytes (21 words) - 04:57, 18 April 2010
- ...e chain bonds to both the alpha carbon and the backbone nitrogen atom, the nitrogen is a secondary amine, making proline an ''imino acid'' rather than an ''ami1 KB (231 words) - 08:46, 8 June 2009
- | pagename = Nitrogen mustards | abc = Nitrogen mustards2 KB (228 words) - 17:05, 22 June 2009
- |{{Image|Terra Nitrogen Plant.jpg|right|200px|Terra Nitrogen plant in Verdigris, Oklahoma that produces ammonia and ammonia derived fert2 KB (246 words) - 22:43, 18 June 2011
- ...t part of the [[flue gas]] from most fossil fuel combustion is uncombusted nitrogen. The next largest part of the flue gas is [[carbon dioxide]] (CO<sub>2</sub ...xides are derived from the nitrogen in the ambient air as well as from any nitrogen-containing compounds in the fossil fuel. The sulfur dioxide is derived from4 KB (619 words) - 09:16, 6 March 2024
- | pagename = Nitrogen mustard | abc = Nitrogen mustard2 KB (231 words) - 04:31, 28 April 2009
- '''Mechlorethamine''' is a specific member of the class of [[nitrogen mustard]]s, originally introduced as [[chemical weapon]]s but now used medi670 bytes (83 words) - 20:14, 1 July 2010
- ...[extremophile]] microbes and invertebrates can survive storage in [[liquid nitrogen]].688 bytes (94 words) - 18:31, 29 December 2020
- ...cyclophosphamide]] is a [[medication]] that is "precursor of an alkylating nitrogen mustard [[antineoplastic agent|antineoplastic ]] and [[immunosuppressive ag557 bytes (72 words) - 03:02, 7 October 2013
- {{r|Nitrogen}}306 bytes (37 words) - 19:38, 31 May 2010
- |{{Image|Terra Nitrogen Plant.jpg|right|200px|Terra Nitrogen plant in Verdigris, Oklahoma that produces ammonia and ammonia derived fert2 KB (273 words) - 17:50, 17 June 2011
- ...ha</math>-amino nitrogen atom from the glutamate <math>\alpha</math>-amino nitrogen by transamination. Glutamate is a major [[neurotransmitter]] in the brain. ...utamate and ammonium ion, with energy derived from the hydrolysis of ATP. Nitrogen metabolism is largely controlled by glutamine synthetase and [[glutamate de3 KB (430 words) - 08:08, 8 June 2009
- {{r|Nitrogen mustards}}458 bytes (58 words) - 20:58, 11 January 2010
- ...s used to produce the useful substance [[ammonia]] from [[hydrogen]] and [[nitrogen]]. ==Sources of hydrogen and nitrogen==7 KB (1,067 words) - 10:08, 28 February 2024
- * Nitrogen in the form of ammonia. ...ounds in the cell, an thus glutamate formation is a major contributor to [[nitrogen assimilation]] in many organisms3 KB (509 words) - 02:33, 8 June 2009
- Auto-populated based on [[Special:WhatLinksHere/Nitrogen mustards]]. Needs checking by a human.525 bytes (65 words) - 19:02, 11 January 2010
- ...n [[Lead|lead]] without burning it or take a mouthful of liquid [[Nitrogen|nitrogen]] (without breathing it in, of course).2 KB (319 words) - 08:34, 6 March 2024
- {{r|Blood urea nitrogen}}680 bytes (81 words) - 00:02, 5 January 2010
- {{r|Nitrogen}}624 bytes (83 words) - 11:58, 31 December 2022
- | pagename = Nitrogen cycle | abc = Nitrogen cycle2 KB (291 words) - 10:53, 26 May 2010
- ...rogen out of soil, and decreased nitrogen itself will reduce plant growth, nitrogen fertilizers are often added to the soil in order to counteract that general2 KB (380 words) - 05:28, 3 January 2008
- In tradition agriculture, [[maize]] (corn) was planted (often with some high-nitrogen fertilizer) such as a small fish under the seeds. Besides its own productio727 bytes (110 words) - 13:41, 29 June 2008
- *{{cite journal|author=Jessica G. Davis|title=The Nitrogen Fate and Transformations Game|journal=Journal of Natural Resources and Life574 bytes (82 words) - 22:02, 11 March 2010
- {{r|Nitrogen mustards}}698 bytes (86 words) - 15:48, 11 January 2010
- ...g a base structure (C<sub>1</sub>N<sub>4</sub>H<sub>2</sub>) in which four nitrogen atoms and one carbon atom form a 5-atom heterocycle. They can be synthesiz672 bytes (100 words) - 14:38, 6 February 2010
- {{r|Nitrogen mustards}}835 bytes (102 words) - 17:24, 11 January 2010
- {{r|Nitrogen}}802 bytes (101 words) - 12:57, 15 March 2024
- ...e of physical state, so that, for example, liquid [[nitrogen]] and gaseous nitrogen do not qualify as allotropes of that element.2 KB (333 words) - 21:17, 13 November 2010
- ...tic]] ring structure with two heteroatoms, at least one of which must be a nitrogen atom. One, and only one, lone pair of electrons from each heteroatom is pa ...r to pyrazole and imadazole shown above, but they would have an additional nitrogen atom in the ring structure.2 KB (340 words) - 08:40, 22 August 2013
- {{r|Nitrogen}}777 bytes (99 words) - 16:15, 11 January 2010
- *{{cite book|author=Gary Maxwell|title=Synthetic Nitrogen Products: A Practical Guide to the Products and Processes|edition=First Edi679 bytes (98 words) - 21:41, 25 February 2008
- {{r|Nitrogen mustard}}703 bytes (107 words) - 19:19, 23 November 2010
- ...a</math>-lactam structure. The lactams may have substitutions added to the nitrogen atom or any of the non-carbonyl carbon atoms in the base structure.2 KB (362 words) - 17:14, 21 March 2024
- ...e.html Eight molecular manipulable models of different compounds of solely nitrogen and oxygen, illustrating the law of multiple proportions.] USC Department o1,010 bytes (128 words) - 21:16, 4 June 2009
- {{r|Nitrogen}}768 bytes (104 words) - 14:34, 10 September 2011
- |molformula= [[Carbon|C]]<sub>9</sub>[[Hydrogen|H]]<sub>13</sub>[[Nitrogen|N]]<sub>4</sub> |molformula= [[Carbon|C]]<sub>9</sub>[[Hydrogen|H]]<sub>13</sub>[[Nitrogen|N]]<sub>4</sub>3 KB (338 words) - 19:37, 22 July 2009
- ...to cyclic aromatic compounds, and their alkyl derivatives, that contain [[nitrogen]], [[oxygen]] or [[sulphur]] atoms within the ringed structure. These cont1 KB (129 words) - 09:16, 6 March 2024
- ...was ©OO. Nitrous oxide (N20) was ØØO, and ØO was nitric oxide (NO), while nitrogen peroxide (NO2) could be represented as ØOO. He had discovered another fund3 KB (538 words) - 21:11, 4 June 2009
- ...rogen-Emissions Sour Showers: Acid Rain Returns--This Time It Is Caused by Nitrogen Emissions]. ''Scientific American'', September 2010, pp. 23-24.1 KB (147 words) - 13:22, 30 October 2023
- A '''nitrile''' is an organic compound containing a carbon triple bonded to nitrogen.827 bytes (113 words) - 17:46, 28 October 2010
- ...th blue light receptors and other environmental stimuli such as carbon and nitrogen sources. The primary models we are using include Arabidopsis thaliana, Cer942 bytes (124 words) - 04:48, 22 November 2023
- ===Nitrogen and hydrogen required for ammonia synthesis=== As another example, if we want to determine the amounts of [[nitrogen]] and hydrogen gases required to produce 5 [[kilogram|kg]] of [[ammonia]] g8 KB (1,289 words) - 22:35, 20 June 2010