Pyrroloquinoline quinone
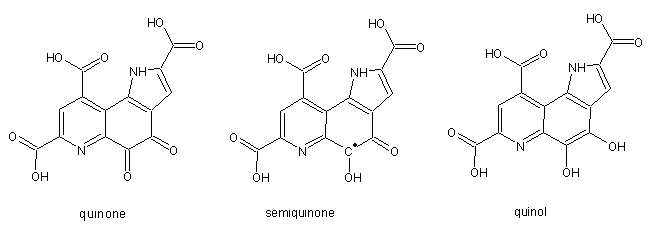
Pyrroloquinoline quinone (PQQ) was originally discovered in 1978 in methanol dehydrogenase fom Hyphomicrobium X. It is present as a non-covalently bound cofactor in several bacterial dehydrogenases involved in the oxidation of alcohols and sugars. The biochemically relevant redox states are the fully oxidized quinone form, the radical semiquinone form and the reduced quinol. The reduced form is only stable when bound to enzyme or at low pH, whereas the semiquinone is only stable at high pH and anerobic conditions.
The pKa values of the protonable groups in PQQ are:
- 0.3 (pyridyl nitrogen)
- 1.6 and 2.2 for the carboxylic groups in the pyridine ring
- 3.3 for the carboxyl group in the pyrrole ring
- 10.3 for the pyrrole nitrogen
PQQ was reported to be a vitamin in mice on April 24, 2003 by a research team led by Takafumi Kato of the Japanese Institute of Physical and Chemical Research (source). However, these conclusions have been strongly challenged.