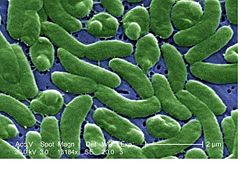
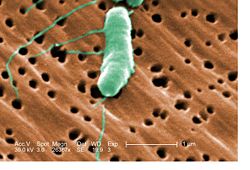
Vibrio vulnificus
| Scientific classification | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||
| Binomial name | ||||||||||||||
| Vibrio vulnificus |
Vibrio vulnificus is a rod-shaped, Gram-negative bacterium that ferments lactose. It is a halophile and can be found in warm coastal waters and is very common in the Gulf of Mexico and its coast. Vibrio vulnificus grows best in the warm months when the water temperature is between 20 to 24° C and cannot be found in cold water.[1]
It is also motile and like two other bacteria found in the Family Vibrionaceae, Vibrio cholerae and Vibrio parahaemolyticus, is a human pathogen. It is known to cause very serious problems and sometimes death in humans if it is ingested or if it contaminates an open wound in the skin.[2]
Strains of V. vulnificus can be divided in three biotypes; strains virulent for humans are categorized in biotypes 1 and 3 while strains that are virulent for fish are classified as biotype 2. Gene sequencing has allowed for the identification of the virulence genes in V. vulnificus and the ability to better the examination of an infection. This organism was first described as a pathogen in 1976 by Hollis et al.
Genome structure
The biotype 1 strain of Vibrio vulnificus has two circular chromosomes and a smaller plasmid. The larger of the two chromosomes is 3,354,505 bp long while the smaller chromosome is 1,857,073 bp long. The plasmid identified was of 48,508 bp. A super- integron containing 188 gene cassettes was also identified. These gene cassettes vary greatly among the different strains and are distinctive for each Vibrio species. Genes that encode for the production of capsular polysaccharide, RTX, toxins, and type IV pilus (all of which play an important role in the virulence of the strain) along with other genes can be found on the chromosome. Multiple rearrangements, gene duplications, and signs of horizontal transfer were also present on the genome.[3]
Cell structure and metabolism
This bacterium can have a flagellum which allows for its motility. It can also have type IV pili which may be a factor of virulence since they help the bacteria attach to the epithelial cells. Some strains of V. vulnificus are capsulated while others are not. The presence of capsular polysaccharide can greatly increase the virulence of the bacteria. Some strains of V. vulnificus are known to ferment lactose while others ferment sucrose.
Ecology
Marine environments and its inhabitants can serve as shelters for V. vulnificus. Organisms that harbor this bacterium include oysters, eels, shellfish and seaweed. When these organisms are consumed either raw or undercooked by humans the V. vulnificus (present in the seafood) can cause great damage to the health of person and sometimes even death.
Pathology
Vibrio vulnificus can cause serious damage to human beings especially since its infectious dose is not known. It can enter the body if one consumes raw or undercooked seafood (especially oysters)or if a preexisting open wound is exposed to contaminated water.
In a healthy person, ingesting V. vulnificus can cause gastroenteritis which is characterized by diarrhea, vomiting, and stomach aches. Sepsis, ulceration, and necrotizing fasciitis among other things are also common if an open wound is infected. In a person who already has a disease that compromises his/her immune system (such as liver disease, cancer, HIV, etc) the consequences of infection by this bacterium are far more severe. V. vulnificus can cause primary septicemia or bloodstream infections which can then lead to high fevers, septic shock and death. There is a fifty percent chance of fatality in immunocompromised persons.
There are a number of enzymes produced by the bacterium that assist in the infection. Some of these enzymes are lipase, DNAase, and mucinase. The presence of polysaccharide capsules, lipopolysaccharide (LPS), RTX toxins and type IV pili (which allow the bacterium to attach to the epithelial cells) allow for a stain to be virulent. People with Vibrio vulnificus infections are treated antimicrobial agents. Some antibiotics that work the most effectively are tetracycline, aminoglycosides, and third- generation cephalosporins.
Application to Biotechnology
This organism does not produce any enzymes that prove beneficial to humans. It does, however, produce endotoxins and enzymes that could increase its virulence. These enzymes and toxins include RTX toxins, lipopolysaccharides, lipases, and proteases among others.
Current Research
Vibrio vulnificus Damages Macrophages during the Early Phase of Infection
Not much is known about the mechanisms through which V. vulnificus causes disease. The pathogenicities of a clinical isolate and of an environmental isolate were tested during this research. The clinical isolate, strain M2799, was described a hundred times more lethal in mice than strain JCM3731, which is the environmental isolate.
Even though there are a number of virulence factors, the expression of capsular polysaccharide (CPS) is related to the fatality in animal models. Strains of V. vulnificus that are encapsulated are much more lethal than strains that are not. This is because the capsular polysaccharide serves to protect the strain from undergoing phagocytosis by macrophages and neutrophils. Studies have shown that lymphocytes in the host are lost through apoptosis induced by V.vulnificus. Through this research it is shown that the clinical strain was very effective in inducing apoptosis in macrophage while the environmental strain was not able to.
There was a high level of cytotoxin activity in strain M2799 which then led to a decrease in the number of macrophages. In strain JCM3731, the cytotoxin level was about thousand fold lower and the macrophages were not affected. The effect of macrophages and neutrophils on the growth of V. vulnificus was also examined in this research. Results show that macrophage function to completely inhibit the growth of strain M2799 and that neutrophils are half as effective in inhibiting the growth of this strain. These results show that macrophages are critical in stopping an infection caused by V. vulnificus and that neutrophils alone cannot prevent the infection but rather only help the macrophage in inhibiting the growth of this bacterium.[4]
RpoS-Dependent Stress Response and Exoenzyme Production in Vibrio vulnificus
This research shows that the presence of the alternate sigma factor, RpoS, plays a very important role in the survival of Vibrio vulnificus. Since this bacterium is exposed to different environments (human hosts along with hosts in the aquatic environment), it must adapt to the stress that these environments cause. A low pH, osmotic stress, starvation, and temperatures that are not suitable for the growth of this bacterium are just some of the stresses. Not only does RpoS help Vibrio vulnificus respond to stressful environmental changes but it is also involved in the organism’s virulence.
To study the role of RpoS in V. vulnificus, researchers created an rpoS mutant strain. This was done by homologous recombination. The results showed that the rpoS mutant was more vulnerable to stress caused by environmental changes, had reduced motility and had a decrease in the activity of five of the nine exoenzymes tested.
Studies have also shown that RpoS regulates the expression of certain genes that encode for proteins involved in tissue damage that arises with the infection. This research demonstrates the importance of RpoS in the survival and virulence of pathogens such as V. vulnificus.[5]
Evidence for an Intermediate Colony Morphology of Vibrio vulnificus
Colonies of Vibrio vulnificus can either be opaque or translucent depending on the presence or absence of a capsular polysaccharide. When the capsular polysaccharide (CPS) is expressed, the colony is known as opaque. If the CPS expression is lost the colony becomes translucent. Researchers studied five strains of V. vulnificus in this experiment.
The results from this research show that there is a strain of intermediate colony morphotype that is not completely translucent and not quite as opaque. Under certain circumstances opaque colonies can switch to translucent colonies. This is allowed by a gene known as wzb. Tests have shown that the strains that do contain the wzb appear as opaque colonies while strains that don’t have this gene form translucent colonies. Intermediate strains do have the wzb gene but reverse transcriptase studies show that the transcription of this gene is reduced. Intermediate colonies are also known to turn into opaque colonies.
In order to figure out whether the intermediate colonies arose from a colony that contained both the opaque and the translucent cells, researchers diluted and plated a few intermediate colonies onto heart infusion (HI) agar plates. If intermediate colonies were a result of a mixture of the opaque and translucent cells, there would be no or a few intermediate colonies growing on the HI agar. This, however, was not the case because the colonies that did grow on the HI agar were of the intermediate morphotype.
The ability of intermediate colonies to turn into translucent colonies was also tested. A specific strain, JY1305, was used for this part of the experiment because this strain makes it easier to differentiate between the intermediate and translucent colonies. Observations showed that the opaque colonies gave rise to both the translucent and the intermediate colonies at a moderate rate while the intermediate strains gave rise to translucent colonies at a much higher rate. The results suggest that the decrease in the capsular polysaccharide on the cell surface can cause the strain to turn into an intermediate morphotype. Studies show that two mechanisms can cause the decrease of the capsular polysaccharide on cellular surface; the absence of the wzb gene is one cause while the down regulation of CPS genes under specific conditions is another reason as to why there is a decrease in CPS and why the colonies are less opaque.[6]
References
- ↑ Todar, Kenneth. Vibrio vulnificus. Dept. of Bacteriology, University of Wisconsin-Madison. 30 March 2008
- ↑ "Vibrio vulnificus General Information." March 27 2008.
- ↑ "Vibrio vulnificus".ed. Yambao, Kristine. 11 July 2007
- ↑ Tsuchiya, T., Mitsuo, E., Hayashi, N., Hikita, Y., Nakao, H., Yamamoto, S., Miyamoto, K., and Tsujibo, H. (2007) Vibrio vulnificus Damages Macrophages during the Early Phase of Infection. Infection and Immunity. Volume 75, pages 4592–6 doi: 10.1128/IAI.00481-07. PMCID: PMC1951150
- ↑ Hülsmann, A., Rosche, T.M., Kong, I.S., Hassan, H.M., Beam, D.M., and Oliver, J.D. (2003)RpoS-Dependent Stress Response and Exoenzyme Production in Vibrio vulnificus. Applied and Environmental Microbiology. Volume 69, pages 6114–20 doi: 10.1128/AEM. 69.10.6114-6120 PMCID: PMC201245
- ↑ Rosche, T.M., Smith, B., and Oliver, J.D. (2006) Evidence for an Intermediate Colony Morphology of Vibrio vulnificus. Applied and Environmental Microbiology. Volume 72, pages 4356–9 doi: 10.1128/AEM.02937-05. PMCID: PMC1489594