Antibiotic
Antibiotics reduce the growth or reproduction of cells, usually bacterial, and are used as medications to treat infections and some cancers. They interfere with the life cycle of cells in a number of different ways. Some antibiotics, like penicillin, interfere with cell wall synthesis.
Antivirals may be reverse transcriptase inhibitors that interefere with the production of viral RNA and DNA. Other antibiotics are nucleoside analogs that get incorporated into the viral RNA or DNA and act a chain terminators.
Classes of antibiotics
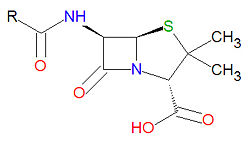
Penicillins
Penicillins have a common beta-lactam base structure, as shown, where R represents different chemical groups. Penicillins work by binding to penicillin-binding proteins irreversibly in a ring-opening reaction and disrupting bacterial cell wall synthesis. Some bacteria are resistant to penicillin because they have acquired the ability to make penicillinases, enzymes which degrade penicillin.
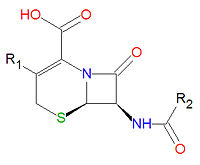
Cephalosporins
Cephalosporins are a class of antibiotic compounds sharing a common beta-lactam base structure, 7-aminocephalosporanic acid (7-ACA), that was derived from the first cephalosporin discovered, cephalosporin C. Penicillins are very similar, although they contain a five-membered ring in place of the six-membered ring present in the cephalosporin. The activity of cephalosporins, penicillins, and some other antibiotics are due to the presence of a beta-lactam, which binds irreversibly, via acylation, to penicillin-binding proteins, thereby inhibiting the peptidogycan layer of bacterial cell wall synthesis. Cephalosporins are often made semisynthetically. Cephalosporins and the very closely relatedcephamycins are collectively referred to as cephems. In general, second generation and later cephalosporins have a broader spectrum of activity against Gram-negative bacteria.
Because the original cephalosporins used the "ceph" form of the spelling and were often trademarked, the International Nonproprietary Names (INN) suggested by the World Health Organization use the "cef" spelling for the generic drug name of all cephalosporins.
|
|
Miscellaneous beta-lactams
Several antibiotics contain the beta-lactam ring, but are not considered either penicillins or cephalosporins.
Tetracyclines
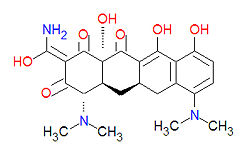
Tetracyclines are antibiotics having a common base structure consisting of four rings conjoined in a linear fashion, with differing chemical groups attached to it, typically on the bottom side or the amino group on the left side in the figure shown. Tetracyclines hinder translation by binding to the 30S ribosomal subunit and preventing the amino-acyl tRNA from binding to the A site of the ribosome, thus disrupting the synthesis of bacterial proteins.
|
|
|
Quinolones
The mechanism of action for quinolones is different from that of macrolides, beta-lactams, aminoglycosides, or tetracyclines, so organisisms resistant to those classes of antibiotic drugs may be susceptible to quinolones. In particular, the quinolones interfere with topoisomerase enzymes, including topoisomerase II (DNA gyrase) and topoisomerase IV, which are vital to bacterial DNA replication, transcription, repair and recombination. Because the use of fluoroquinolones may lead to tendinitis or tendon rupture, especially in the Achilles tendon, the FDA requires a "black box" warning for these medications. The cause of the tendon damage is not yet determined.
|
|
Aminoglycosides
All patients taking aminoglycoside antibiotics should be under close observation due to concerns of ototoxicity and nephrotoxicity. These antibiotics have low activity against Gram-positive bacteria and are often used in conjuntion with other antibiotics from a different antibiotic class. They function by inhibiting bacterial protein synthesis.
Macrolides and ketolides
Macrolide antibiotics function by binding to the 50S subunit of the bacterial 70S ribosome, thus interferring with the translocation of peptides and the production of bacterial proteins.
- Azithromycin (an azalide, which is a subclass of macrolides)
- Clarithromycin
- Dirithromycin
- Erythromycin
- Roxithromycin
- Telithromycin (a ketolide)
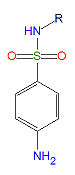
Sulfonamides
Sulfonamides, (R-SO2-NH2) are competitive inhibitors of para-aminobenzoic acid (PABA), the natural substrate for the enzymedihydropteroate synthetase, which is required within the folic acid cycle for the production of folic acid. The sulfonamides are bacteriostatic rather than bacteriocidal. Bacterial resistance to one sulfonamide indicates resistance to all of them.
Glycopeptides
- Bleomycin used as for cancer chemotherapy, not as an antibacterial
- Ramoplanin
- Teicoplanin
- Vancomycin
Oxazolidinones
Other Antibiotic
- Chloramphenicol
- Clindamycin
- Colistin
- Fosfomycin
- Loracarbef
- Metronidazole
- Nalidixic Acid
- Nitrofurantoin
- Polymyxin B Sulfate
- Procaine
- Spectinomycin
- Tinidazole
- Trimethoprim
Adverse effects
Antibiotic resistance
A number of organisms have developed resistance to antibiotics. At the microbial level, this may be due either to the antibiotic therapy allowing the survival of naturally resistant organisms of the species, or of the transfer of resistance genetic factors among bacteria.
Diarrhea
Antibiotic associated diarrhea sometimes includes pseudomembranous enterocolitis caused by Clostridium difficile. Antibiotic associated diarrhea may be prevented by administering probiotics such as Lactobacillus.[1]
References
- ↑ Kale-Pradhan PB, Jassal HK, Wilhelm SM (2010). "Role of Lactobacillus in the prevention of antibiotic-associated diarrhea: a meta-analysis.". Pharmacotherapy 30 (2): 119-26. DOI:10.1592/phco.30.2.119. PMID 20099986. Research Blogging.
Primary references
The most up-to-date information about Antibiotic and other drugs can be found at the following sites.
- Antibiotic - FDA approved drug information (drug label) from DailyMed (U.S. National Library of Medicine).
- Antibiotic - Drug information for consumers from MedlinePlus (U.S. National Library of Medicine).
- Antibiotic - Detailed information from DrugBank.
- Pages using PMID magic links
- CZ Live
- Health Sciences Workgroup
- Chemistry Workgroup
- Infectious Disease Subgroup
- Pharmacology Subgroup
- Biochemistry Subgroup
- Articles written in American English
- Advanced Articles written in American English
- All Content
- Health Sciences Content
- Chemistry Content
- Infectious Disease tag
- Pharmacology tag
- Reviewed Passed