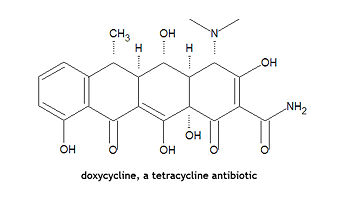
Doxycycline
Doxycycline, is "a synthetic tetracycline derivative with similar antimicrobial activity. Animal studies suggest that it may cause less tooth staining than other tetracyclines. It is used in some areas for the treatment of chloroquine-resistant falciparum malaria".[1] Doxycycline can also be take prophylactically in the prevention of malaria. It can be used to treat anthrax, brucellosis, cholera, ornithosis, plague, tularemia, rickettsioses. Its chemical name is (4S,4aR,5S,5aR,6R,12aS)-4-(dimethylamino)-1,4,4a,5,5a,6,11,12a-octahydro-3,5,10,12,12a-pentahydroxy-6-methyl-1,11-dioxo-2-naphthacenecarboxamide.
Pharmacokinetics
Absorption
Immediate-release formulations of doxycycline, similar to tetracycline[2], has absorption reduced or delayed by milk.[3] However, the milk interaction is much smaller for doxycycline and of uncertain importance.[4]
Distribution
Metabolism
Excretion
Medical uses
References
- ↑ National Library of Medicine. Doxycycline. Retrieved on 2007-11-08.
- ↑ Leyden JJ (1985). "Absorption of minocycline hydrochloride and tetracycline hydrochloride. Effect of food, milk, and iron". J. Am. Acad. Dermatol. 12 (2 Pt 1): 308–12. PMID 3838321. [e]
- ↑ Meyer FP, Specht H, Quednow B, Walther H (1989). "Influence of milk on the bioavailability of doxycycline--new aspects". Infection 17 (4): 245–6. PMID 2767766. [e]
- ↑ Welling PG, Koch PA, Lau CC, Craig WA (1977). "Bioavailability of tetracycline and doxycycline in fasted and nonfasted subjects". Antimicrob. Agents Chemother. 11 (3): 462–9. PMID 856000. [e]
External links
The most up-to-date information about Doxycycline and other drugs can be found at the following sites.
- Doxycycline - FDA approved drug information (drug label) from DailyMed (U.S. National Library of Medicine).
- Doxycycline - Drug information for consumers from MedlinePlus (U.S. National Library of Medicine).
- Doxycycline - Detailed information from DrugBank.