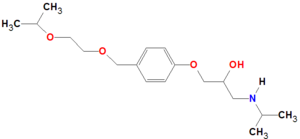
Bisoprolol
Jump to navigation
Jump to search

|
| |||||||
| Bisoprolol | |||||||
| |||||||
| Uses: | adrenergic beta-antagonist | ||||||
| Properties: | |||||||
| Hazards: | |||||||
| |||||||
In medicine, bisoprolol is a "cardioselective beta-1-adrenergic blocker. It is effective in the management of hypertension and angina pectoris."[1]
Variation in the effects of bisoprolol may be due to adrenergic receptor genotypes.[2]
History
Bisoprolol fumarate was approved in 1992.[3] Generic bisoprolol was approved in 2002.[4]
Metabolism
It is not metabolized by cytochrome P-450 2D6 allele.
Dosage
For healthy adults, the starting dose is 5 mg orally per day (sometimes 2.5 mg) and the maximum dose is 10 to 20 mg orally per day.
Synoyms and Brand Names
Synonyms
- bisoprolol fumarate
- bisoprolol hemifumarate
- bisoprolol fumerate
Brand Names
- Cardicor
- Concor
- Condyline
- Condylox
- Detensiel
- Emconcor
- Emcor
- Euradal
- Isoten
- Monocor
- Soloc
- Soprol
- Zebeta
External links
The most up-to-date information about Bisoprolol and other drugs can be found at the following sites.
- Bisoprolol - FDA approved drug information (drug label) from DailyMed (U.S. National Library of Medicine).
- Bisoprolol - Drug information for consumers from MedlinePlus (U.S. National Library of Medicine).
- Bisoprolol - Detailed information from DrugBank.
References
- ↑ Anonymous (2024), Bisoprolol (English). Medical Subject Headings. U.S. National Library of Medicine.
- ↑ Zaugg M, Bestmann L, Wacker J, et al (July 2007). "Adrenergic receptor genotype but not perioperative bisoprolol therapy may determine cardiovascular outcome in at-risk patients undergoing surgery with spinal block: the Swiss Beta Blocker in Spinal Anesthesia (BBSA) study: a double-blinded, placebo-controlled, multicenter trial with 1-year follow-up". Anesthesiology 107 (1): 33–44. DOI:10.1097/01.anes.0000267530.62344.a4. PMID 17585213. Research Blogging.
- ↑ Drugs@FDA. U S Food and Drug Administration
- ↑ Drugs@FDA. U S Food and Drug Administration