Clostridium difficile
| Clostridium difficile | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Scientific classification | ||||||||||||||
| ||||||||||||||
| Binomial name | ||||||||||||||
| Clostridium difficile |
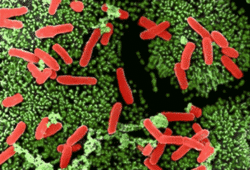
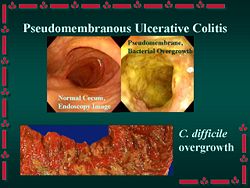
Clostridium difficile is a spore-forming, anaerobic, toxin-producing bacterium that is a "common inhabitant of the colon flora in human infants and sometimes in adults. It produces a toxin that causes pseudomembranous enterocolitis in patients receiving antibiotic therapy."[1] C. difficile superinfection after oral antibiotic therapy, leading to potentially fatal pseudomembranous enterocolitis, has been an increasingly severe public health problem. Indeed, many primary physicians now consider it wise to warn outpatients on antibiotics to seek immediate consultation if they develop severe diarrhea.
C. difficile is present at low levels in the gut flora of about 3% of adults. These people however show no symptoms and do not need to be treated. The infection occurs when a person is treated with antibiotics targeted against other bacteria. The disease is for the most part nosocomial. Patients who are hospitalized come in contact and are often inoculated with the bacteria. When the patient is treated with antibiotics, especially those with a broad range of activity, the normal gut flora is disrupted, and C. difficile, with its multi-drug resistance, experiences overgrowth. The bacteria releases large quantities of enterotoxins (toxin A) and cytotoxins (toxin B), causing pseudomembranous enterocolitis.
History
In 1935, Hall and O’Toole first isolated the bacteria from the stools of newborns and described it. They named it Bacillus difficilis because it was hard to isolate and grew very slowly in culture.[2]
C. difficile is an important pathogen that is currently increasing in its prevalence world-wide. A complete genome sequence would enable geneticists to come up with a more direct and efficient treatment against the pathogen. The genetic material encodes for antimicrobial resistance, production of toxins (virulence), host interaction (adaptations for survival and growth within the gut environment), and the production of surface structures. The understanding of how these genes interact with their environment will be useful in developing therapies against C. difficile associated diseases.[3]
Genome Structure
Sebaihia et al (2006) determined the complete genomic sequence of C. difficile strain 630, a highly virulent and multidrug-resistant strain. It was found that the genome consists of a circular chromosome of 4,290,252 bp and a plasmid, pCD630, of 7,881 bp. The chromosome encodes 3,776 predicted coding sequences (CDSs), with resistance, virulence, and host interaction genes, while the plasmid carries only 11 CDSs, none of which has any obvious function. C. difficile has a highly mobile genome, with 11% of the genome consisting of mobile genetic elements, mostly in the form of conjugative transposons. Conjugative transposons are mobile genetic elements that are capable of integrating into and excising from the host genome and transferring themselves, and are responsible for the evolutionary acquisition by C. difficile of genes involved in resistance, virulence, and host interactions. Some of the mobile elements are prophage sequences. Host interaction genes involve genes that code for metabolic capability adaptations for survival and growth within the gut environment.[3]
The genome carries several copies of a very interesting genetic element, the IStron. The IStron is a hybrid between an intron and an insertion sequence (IS), and can therefore insert itself into a DNA sequence, and later be excised from the primary mRNA transcript by cellular machinery. For example, a copy inserted in the tcdA gene (that encodes the enterotoxin A) renders the gene nonfunctional until the IStron is excised. Truncated copies of the IStron (containing only the IS sequence) were found in intergenic regions. Possibly, these truncated variants may be the answer to make C. difficile unable to produce functional toxins. [3]
Cell structure and metabolism
Clostridia are Gram-positive, anaerobic, spore-forming, rod-shaped, motile bacteria. As spores, the bacteria look like drumsticks, with a bulge located at one end. C. difficile produces an S layer (polysaccharide capsule) that contributes to its pathogenicity. C. difficile has flagellae that contribute to its motility.[4]
C. difficile is an obligate fermenter, and exhibits optimal growth in blood agar at human body temperatures in the absence of oxygen. The metabolism of C. difficile is in large part adapted to life in the intestinal tract.[4] It produces enzymes that degrade nutrients abundant in the intestine. Carbohydrates are the preferred nutrient source, and C. difficile has the ability to metabolize a wide range of carbohydrates. The bacteria also have a relatively unique ability to utilize ethanolamine, an abundant phospholipid provided by the host’s dietary intake, as a carbon and nitrogen source.[3]
C. difficile has the enzyme that catalyzes the decarboxylation of p-hydroxyphenylacetate (a tyrosine degradation product) to p-crysol, a compound that stunts bacterial growth. C. difficile produces and tolerates high concentrations of p-crysol, giving it a competitive advantage over the normal bacterial flora in the intestine.[3] '
Ecology
C. difficile is found throughout nature, especially in soil. It exhibits optimal growth in blood agar at human body temperatures in the absence of oxygen. It can remain dormant in hospitals in the form of spores until patient inoculation. Following treatment of antibiotics, C. difficile overgrows in the intestinal tract, causing pseudomembranous enterocolitis, and consequently diarrhea.[4]
Pathology
C. difficile is transmitted from person to person by the fecal-oral route in the form of vegetative cells and heat-resistant spores. Most vegetative cells are killed in the stomach, but spores pass through the stomach unaffected because of their acid resistance. Spores germinate in the small bowel upon exposure to bile acids. C. difficile multiplies in the colon and overgrows in the absence of competitors (following antibiotic treatment). Mucosa of the gut allows C. difficile adherence to the colonic epithelium.[5]
Different pathogenic strains of C. difficile produce different toxins, but the most common ones are enterotoxin (toxin A) and cytotoxin (toxin B). The two toxins lead to "the production of tumour necrosis factor-alpha and pro-inflammatory interleukins, increased vascular permeability, neutrophil and monocyte recruitment, opening of epithelial cell junctions and epithelial cell apoptosis." Hydrolytic enzymes cause connective tissue to degrade, leading to colitis, pseudomembrane formation and watery diarrhea.[5] Toxin B is thought to be essential for disease to occur.[6]
Risk factors for acquisition include use of proton pump inhibitor medications.[7][8][9]
Clostridium difficile infection (CDI) can range in severity from asymptomatic to life threatening. Death is more common among the aged (>65). Patient symptoms include severe diarrhea, exposure to antibiotics, foul stool odor, and abdominal pain.[5]
Diagnosis
Clinical practice guidelines address diagnosis.[10]
Two clinical prediction rules suggests which patients will have positive toxin results.[11][12] Unfortunately, neither of these studies used the sensitive polymerase chain reaction as a reference standard.
- Clostridium difficile is very unlikely among patients "without a history of antibiotic use and either significant diarrhea or abdominal pain."[11]
Suggestive medical history
A history of exposure to antibiotics in the last 30 days is present in most all patients.[11]
Clostridium difficile should be suspected among patients with "clinically significant diarrhea, usually defined as 3 or more loose stools per day for at least 1 to 2 days".[13] Significant diarrhea is defined as one day of "at least three partially formed or watery stools, 1 L of colostomy output, or more than 200 ml of watery rectal bad output" or "multiple or several loose stools."[11]
Laboratory tests
The accuracy of laboratory tests is in the table.[14]
| Sensitivity | Specificity | |
|---|---|---|
| Cytotoxin by enzyme immunoassay | 73% | 97% |
| Cell culture cytotoxin assay (tissue culture assay) |
77% | 97% |
| Real-time polymerase chain reaction for toxin B gene |
93% | 97% |
| Anaerobic culture (for toxigenic C. difficile strains) |
100% | 96% |
| Sigmoidoscopy[15] (for pseudomembranes) |
31% |
Colonoscopy
Based on a few studies, most patients with Clostridium difficile associated diarrhea do not have pseudomembranous enterocolitis upon colonoscopy.[16][17]
Treatment
Clinical practice guidelines[10] and a systematic review[18] address treatment.
Antibiotics
| Trial | Patients | Intervention | Comparison | Outcome | Results - cure | Results - recurrence | ||
|---|---|---|---|---|---|---|---|---|
| Intervention | Control | Intervention | Control | |||||
| Louie et al[19] 2011 |
629 patients • mild to severe |
Fidaxomicin • 200 mg twice daily • 10 days |
Vancomycin • 125 mg 4 times/day • 10 days |
stool toxin after 4 weeks | 88% | 86% | 15% | 25% |
| Zar et al[20] 2007 |
150 patients • mild to severe |
Metronidazole • 250 mg 4 times/day • 10 days |
Vancomycin • 125 mg 4 times/ day • 10 days |
stool toxin after 3 weeks | 90% (76% if severe) |
98% (97% if severe) |
15% | 14% |
Various antibiotics have been studied in randomized controlled trials.[21][22][23] Teicoplanin may be the most effective antibiotic.[21][22] Vancomycin has an insignificant trend towards being better than metronidazole;[21] metronidazole is likely to be the least expensive of the group and is not a last resort for other highly resistant organisms.
"Our findings suggest that metronidazole and vancomycin are equally effective for the treatment of mild CDAD, but vancomycin is superior for treating patients with severe CDAD." according to a randomized controlled trial. [20]
In a randomized controlled trial, the relative risk ratio of fidaxomicin, as compared to vancomycin, for recurrence of C. difficile infection was 0.6 and the relative risk reduction was 40.0%. In populations similar to those in this study which had a rate of risk as measured by the recurrence of C. difficile infection of 25% without treatment, the number needed to treat is 10. [19]
Consider vancomycin if the patient has two or more points from the following:[24]
- 1 point for each of
- WBC > 15k
- albumin < 2.5
- age > 60
- temperature > 38.3° C
- 2 points for each of
- Pseudomembranous colitis
- Intensive care
It should be noted that vancomycin is given orally in this condition, and is not absorbed systemically. While it still needs to be used with care to avoid the development of resistant organisms, the risk may be lower than if it is used as a parenteral therapy for already resistant pathogens such as methicillin-resistant Staphylococcus aureus.
In a randomized controlled trial, the relative risk ratio of fidaxomicin, as compared to vancomycin, for recurrence of C. difficile infection was 0.6 and the relative risk reduction was 40.0%. In populations similar to those in this study which had a rate of risk as measured by the recurrence of C. difficile infection of 25% without treatment, the number needed to treat is 10. [19]
A case series suggests tigecycline for severe disease.[25]
Probiotics (administration of bacteria)
Probiotic administration may not treat[26]help according to a meta-analyses. Probiotics can be harmful among intensive care patients.[27]
|title=Relapsing clostridium difficile enterocolitis cured by rectal infusion of homologous faeces |journal=Lancet |volume=2 |issue=8354 |pages=845 |year=1983 |month=October |pmid=6137662 |doi= |url=http://linkinghub.elsevier.com/retrieve/pii/S0140-6736(83)90753-5 |issn=}}</ref>[28] or colonoscopic[29] infusion of feces helped according to case reports.
Monoclonal antibodies
In a randomized controlled trial, the relative risk ratio of adjunct treatment with monoclonal antibodies against C. difficile toxins, as compared to placebo, for recurrence of C. difficile infection was 0.3 and the relative risk reduction was 72.0%. In populations similar to those in this study which had a rate of risk as measured by the recurrence of C. difficile infection of 25% without treatment, the number needed to treat is 6. [30]
Recurrent infection
Regarding the cause of recurrences:[31]
- Second episode within 8 weeks, 88% are relapses
- Second episode after > 8 weeks, 65% are relapse
A clinical prediction rule found that recurrent infection is more likely if age is more than 65 years, the patient has severe or fulminant illness, and additional antibiotic exposure occurs after after treatment of the initial Clostridium difficile infection.[32] Use of antacids may also be a risk factor for recurrence.[33]
Asymptomatic carriage should not be treated according to a meta-analysis.[34]
Antibiotics
Observational studies conflict regarding the best agent and suggest vancomycin may[35] or may not[36] be better than metronidazole. Various methods exist for the administration of vancomycin[35][37] and metronidazole[38].
| Patients | Intervention / duration | Comparison | Outcome: Recurrence rate | |
|---|---|---|---|---|
| Approximately 2 weeks | ||||
| Vancomycin constant dose[36] | 171 patients | 0.5 to 2 grams daily 10 - 14 days |
NA | 40% |
| Metronidazole constant dose[36] | 115 patients | 1.0 to 1.5 grams per day 10 - 14 days |
NA | 37% |
| Metronidazole constant dose[38] | 20 patients | Metronidzole 1200 mg daily 14 days |
NA | 45% |
| Vancomycin constant dose[35] | 83 patients | 0.5 to 3 grams/day 10 - 16 days |
NA | 54% |
| high dose[35] | 21 patients | ≥2 grams/day 10 - 16 days |
NA | 43% |
| medium dose[35] | 14 patients | 1 - 2 grams/day 10 - 16 days |
NA | 71% |
| low dose[35] | 48 patients | < 1 grams/day 10 - 16 days |
NA | 54% |
| Metronidazole constant dose[35] | 36 patients | 0.5 to 3 grams/day 10 - 16 days |
NA | 42% |
| Approximately 3 weeks | ||||
| Vancomycin tapered dose[35] | 29 patients | Varying doses 21.5 ± 10.0 days |
NA | 31% |
| Vancomycin pulsed dose[35] | 7 patients | Varying doses 21 days |
NA | 14% |
| More than 3 weeks | ||||
| Vancomycin followed by rifaximin[39] | 8 patients | Vancomycin of unknown duration followed by rifaximin 400–800 mg daily for 14 days ≥ 21 days |
NA | 13% |
| Vancomycin tapered & pulsed dose[37] | 22 patients | 125 mg four times daily tapered to 125 mg every third day 42 days |
NA | 0% |
Serial therapy with vancomycin and rifaximin has been studied in a small uncontrolled series of patients.[39]
Administration of bacteria
Probiotics may help.[40] However, probiotics can be harmful among intensive care patients.[27]
Rectal[41][28] or colonoscopic[29] infusion of feces helped according to case reports.
Prevention
Administration of bacteria
Probiotic administration may prevent C diff diarrhea according to meta-analysis meta-analyses[42][40] and a more recent randomized controlled trial.[43] However, probiotics can be harmful among intensive care patients.[27]
Prognosis
A leukocyte count at least 20.0 109/L or a creatinine level (200 µmol/L or 2.2 mg/dl or greater) predict adverse outcomes.[44]
Research
There is an ongoing search for new therapeutic strategies to battle the ever-increasing pathogenicity of the C. difficile strains. Barreto et al (2008) have recently released a study on the effects of the polysaccharide fucoidin, an L-selectin blocker, on toxin-A-induced mouse enteritis. Enteritis is medical term that refers to the inflammation of the intestine, usually accompanies by diarrhea. Fucoidin or the placebo (saline) was injected into the mouse, and 5 minutes later, toxin A was locally released. To measure the effect of fucoidin, intestinal fluid volume/length and ileal loop weight/length ratios were calculated three hours later. It was found that fucoidin significantly (P < 0.05) reduced the toxin-A-induced increase in weight/length and volume/length ratios and reduced mucosal disruption.[45]
Gerber and Ackermann (2008) have published a report on another treatment option, OPT-80. OPT-80 (otherwise known as tiacumicin B, lipiarmycin or PAR-101) is “an antimicrobial with little or no systemic absorption after oral administration and narrow activity spectrum against Gram-positive aerobic and anaerobic bacteria.” OPT-80 is highly effective at very low concentrations, making it a very safe and efficient drug. More clinical trials are to be completed, but for now results look promising.[46]
The reason why C. difficile-associated disease (CDAD) is becoming such a widespread nosocomial disease is because the spores it forms are resistant to heat, chemicals, antibiotics, and radiation, making the hospital environment difficult to sanitize. The study of C. difficile germination from spores to vegetative cells will shed light on the strengths and weaknesses of the spores, and will thus lead to the development of methods for spore eradication. Sorg and Sonenshein (2008) studied the effects of the components of bile on the germination of C. difficile spores. They found that cholate derivatives and glycine act to induce germination in the duodenum. It was found that, interestingly, deoxycholate, a metabolite of cholate (produced by the normal intestinal flora) induces germination while preventing growth of vegetative cells. This implies a mechanism by which normal gut flora can mediate the levels of vegetative C. difficile cells in the gut.[47]
References
- ↑ Anonymous (2024), term (English). Medical Subject Headings. U.S. National Library of Medicine.
- ↑ Hall I, O'Toole E (1935). "Intestinal flora in newborn infants with a description of a new pathogenic anaerobe, Bacillus difficilis". Am. J. Dis. Child 49: 390.
- ↑ Jump up to: 3.0 3.1 3.2 3.3 3.4 Sebaihia M et al (2006). "The multidrug-resistant human pathogen Clostridium difficile has a highly mobile, mosaic genome". Na. Gen. 38: 779–86. PMID 16804543. [e]
- ↑ Jump up to: 4.0 4.1 4.2 Clostridium difficile. 12 Apr. 2008. Wikipedia, the free encyclopedia. 13 Apr. 2008. <http://en.wikipedia.org/wiki/Clostridium_difficile>
- ↑ Jump up to: 5.0 5.1 5.2 C. difficile. 12 Apr. 2008. The Writing Lab and OWL at Purdue and Purdue University. 10 March 2006. <http://media.romanvenable.net/images/cDiffLarge.jpeg>
- ↑ Lyras D, O'Connor JR, Howarth PM, Sambol SP, Carter GP, Phumoonna T et al. (2009). "Toxin B is essential for virulence of Clostridium difficile.". Nature 458 (7242): 1176-9. DOI:10.1038/nature07822. PMID 19252482. PMC PMC2679968. Research Blogging.
- ↑ Kwok CS, Arthur AK, Anibueze CI, Singh S, Cavallazzi R, Loke YK (2012). "Risk of Clostridium difficile Infection With Acid Suppressing Drugs and Antibiotics: Meta-Analysis.". Am J Gastroenterol 107 (7): 1011-9. DOI:10.1038/ajg.2012.108. PMID 22525304. Research Blogging.
- ↑ Janarthanan S, Ditah I, Adler DG, Ehrinpreis MN (2012). "Clostridium difficile-Associated Diarrhea and Proton Pump Inhibitor Therapy: A Meta-Analysis.". Am J Gastroenterol 107 (7): 1001-10. DOI:10.1038/ajg.2012.179. PMID 22710578. Research Blogging.
- ↑ Howell MD, Novack V, Grgurich P, Soulliard D, Novack L, Pencina M et al. (2010). "Iatrogenic gastric acid suppression and the risk of nosocomial Clostridium difficile infection.". Arch Intern Med 170 (9): 784-90. DOI:10.1001/archinternmed.2010.89. PMID 20458086. Research Blogging.
- ↑ Jump up to: 10.0 10.1 Cohen SH, Gerding DN, Johnson S, Kelly CP, Loo VG, McDonald LC et al. (2010). "Clinical practice guidelines for Clostridium difficile infection in adults: 2010 update by the society for healthcare epidemiology of America (SHEA) and the infectious diseases society of America (IDSA).". Infect Control Hosp Epidemiol 31 (5): 431-55. DOI:10.1086/651706. PMID 20307191. Research Blogging.
- ↑ Jump up to: 11.0 11.1 11.2 11.3 Katz DA, Bates DW, Rittenberg E, et al (January 1997). "Predicting Clostridium difficile stool cytotoxin results in hospitalized patients with diarrhea". J Gen Intern Med 12 (1): 57–62. PMID 9034947. PMC 1497057. [e]
- ↑ Cooper GS, Lederman MM, Salata RA (January 1996). "A predictive model to identify Clostridium difficile toxin in hospitalized patients with diarrhea". Am. J. Gastroenterol. 91 (1): 80–4. PMID 8561149. [e]
- ↑ Peterson LR, Robicsek A (2009). "Does my patient have Clostridium difficile infection?". Ann Intern Med 151 (3): 176-9. PMID 19652187.
- ↑ Jump up to: 14.0 14.1 Peterson LR, Manson RU, Paule SM, et al (November 2007). "Detection of toxigenic Clostridium difficile in stool samples by real-time polymerase chain reaction for the diagnosis of C. difficile-associated diarrhea". Clin. Infect. Dis. 45 (9): 1152–60. DOI:10.1086/522185. PMID 17918076. Research Blogging.
- ↑ Seppala, K, Hjelt, L, Supponen, P. Colonoscopy in the diagnosis of antibiotic-associated colitis. Scand J Gastroenterol 1981; 16:465. PMID 7323683
- ↑ Marts BC, Longo WE, Vernava AM, Kennedy DJ, Daniel GL, Jones I (August 1994). "Patterns and prognosis of Clostridium difficile colitis". Dis. Colon Rectum 37 (8): 837–45. PMID 8055732. [e]
- ↑ Pupaibool J, Khantipong M, Suankratay C (January 2008). "A study of Clostridium difficile-associated disease at King Chulalongkorn Memorial Hospital, Thailand". J Med Assoc Thai 91 (1): 37–43. PMID 18386542. [e]
- ↑ Drekonja DM, Butler M, Macdonald R, Bliss D, Filice GA, Rector TS et al. (2011). "Comparative Effectiveness of Clostridium difficile Treatments: A Systematic Review.". Ann Intern Med 155 (12): 839-47. DOI:10.1059/0003-4819-155-12-201112200-00007. PMID 22184691. Research Blogging.
- ↑ Jump up to: 19.0 19.1 19.2 19.3 Louie TJ, Miller MA, Mullane KM, Weiss K, Lentnek A, Golan Y et al. (2011). "Fidaxomicin versus vancomycin for Clostridium difficile infection.". N Engl J Med 364 (5): 422-31. DOI:10.1056/NEJMoa0910812. PMID 21288078. Research Blogging.
- ↑ Jump up to: 20.0 20.1 20.2 Zar FA, Bakkanagari SR, Moorthi KM, Davis MB (2007). "A comparison of vancomycin and metronidazole for the treatment of Clostridium difficile-associated diarrhea, stratified by disease severity.". Clin Infect Dis 45 (3): 302-7. DOI:10.1086/519265. PMID 17599306. Research Blogging.
- ↑ Jump up to: 21.0 21.1 21.2 Nelson R (2007). "Antibiotic treatment for Clostridium difficile-associated diarrhea in adults". Cochrane Database Syst Rev (3): CD004610. DOI:10.1002/14651858.CD004610.pub3. PMID 17636768. Research Blogging.
- ↑ Jump up to: 22.0 22.1 Wenisch C, Parschalk B, Hasenhündl M, Hirschl AM, Graninger W (May 1996). "Comparison of vancomycin, teicoplanin, metronidazole, and fusidic acid for the treatment of Clostridium difficile-associated diarrhea". Clin. Infect. Dis. 22 (5): 813–8. PMID 8722937. [e]
- ↑ Musher DM, Logan N, Bressler AM, Johnson DP, Rossignol JF (January 2009). "Nitazoxanide versus Vancomycin in Clostridium difficile Infection: A Randomized, Double-Blind Study". Clin. Infect. Dis.. DOI:10.1086/596552. PMID 19133801. Research Blogging.
- ↑ Zar FA, Bakkanagari SR, Moorthi KM, Davis MB. A comparison of vancomycin and metronidazole for the treatment of Clostridium difficile-associated diarrhea, stratified by disease severity. Clin Infect Dis. 2007 Aug 1;45(3):302-7. PMID 17599306
- ↑ Herpers BL, Vlaminckx B, Burkhardt O, et al. (June 2009). "Intravenous tigecycline as adjunctive or alternative therapy for severe refractory Clostridium difficile infection". Clin. Infect. Dis. 48 (12): 1732–5. DOI:10.1086/599224. PMID 19435431. Research Blogging.
- ↑ Pillai A, Nelson R (2008). "Probiotics for treatment of Clostridium difficile-associated colitis in adults". Cochrane Database Syst Rev (1): CD004611. DOI:10.1002/14651858.CD004611.pub2. PMID 18254055. Research Blogging.
- ↑ Jump up to: 27.0 27.1 27.2 Muñoz P, Bouza E, Cuenca-Estrella M, et al (June 2005). "Saccharomyces cerevisiae fungemia: an emerging infectious disease". Clin. Infect. Dis. 40 (11): 1625–34. DOI:10.1086/429916. PMID 15889360. Research Blogging.
- ↑ Jump up to: 28.0 28.1 Nieuwdorp M, van Nood E, Speelman P, et al (August 2008). "[Treatment of recurrent Clostridium difficile-associated diarrhoea with a suspension of donor faeces]" (in Dutch; Flemish). Ned Tijdschr Geneeskd 152 (35): 1927–32. PMID 18808083. [e]
- ↑ Jump up to: 29.0 29.1 Mattila E, Uusitalo-Seppälä R, Wuorela M, Lehtola L, Nurmi H, Ristikankare M et al. (2012). "Fecal transplantation, through colonoscopy, is effective therapy for recurrent Clostridium difficile infection.". Gastroenterology 142 (3): 490-6. DOI:10.1053/j.gastro.2011.11.037. PMID 22155369. Research Blogging.
- ↑ Lowy I, Molrine DC, Leav BA, Blair BM, Baxter R, Gerding DN et al. (2010). "Treatment with monoclonal antibodies against Clostridium difficile toxins.". N Engl J Med 362 (3): 197-205. DOI:10.1056/NEJMoa0907635. PMID 20089970. Research Blogging.
- ↑ Kamboj M, Khosa P, Kaltsas A, Babady NE, Son C, Sepkowitz KA (2011). "Relapse versus reinfection: surveillance of Clostridium difficile infection.". Clin Infect Dis 53 (10): 1003-6. DOI:10.1093/cid/cir643. PMID 21976462. Research Blogging.
- ↑ Hu MY, Katchar K, Kyne L, et al (December 2008). "Prospective Derivation and Validation of a Clinical Prediction Rule for Recurrent Clostridium difficile Infection". Gastroenterology. DOI:10.1053/j.gastro.2008.12.038. PMID 19162027. Research Blogging.
- ↑ Garey KW, Sethi S, Yadav Y, DuPont HL (December 2008). "Meta-analysis to assess risk factors for recurrent Clostridium difficile infection". J. Hosp. Infect. 70 (4): 298–304. DOI:10.1016/j.jhin.2008.08.012. PMID 18951661. Research Blogging.
- ↑ Shim JK, Johnson S, Samore MH, Bliss DZ, Gerding DN (February 1998). "Primary symptomless colonisation by Clostridium difficile and decreased risk of subsequent diarrhoea". Lancet 351 (9103): 633–6. DOI:10.1016/S0140-6736(97)08062-8. PMID 9500319. Research Blogging.
- ↑ Jump up to: 35.0 35.1 35.2 35.3 35.4 35.5 35.6 35.7 35.8 McFarland LV, Elmer GW, Surawicz CM (July 2002). "Breaking the cycle: treatment strategies for 163 cases of recurrent Clostridium difficile disease". Am. J. Gastroenterol. 97 (7): 1769–75. DOI:10.1016/S0002-9270(02)04195-3. PMID 12135033. Research Blogging.
- ↑ Jump up to: 36.0 36.1 36.2 Pépin J, Routhier S, Gagnon S, Brazeau I (March 2006). "Management and outcomes of a first recurrence of Clostridium difficile-associated disease in Quebec, Canada". Clin. Infect. Dis. 42 (6): 758–64. DOI:10.1086/501126. PMID 16477549. Research Blogging.
- ↑ Jump up to: 37.0 37.1 Tedesco FJ, Gordon D, Fortson WC (November 1985). "Approach to patients with multiple relapses of antibiotic-associated pseudomembranous colitis". Am. J. Gastroenterol. 80 (11): 867–8. PMID 4050760. [e]
- ↑ Jump up to: 38.0 38.1 Mattila E, Anttila VJ, Broas M, et al (2008). "A randomized, double-blind study comparing Clostridium difficile immune whey and metronidazole for recurrent Clostridium difficile-associated diarrhoea: efficacy and safety data of a prematurely interrupted trial". Scand. J. Infect. Dis. 40 (9): 702–8. PMID 19086244. [e]
- ↑ Jump up to: 39.0 39.1 Johnson S, Schriever C, Galang M, Kelly CP, Gerding DN (March 2007). "Interruption of recurrent Clostridium difficile-associated diarrhea episodes by serial therapy with vancomycin and rifaximin". Clin. Infect. Dis. 44 (6): 846–8. DOI:10.1086/511870. PMID 17304459. Research Blogging.
- ↑ Jump up to: 40.0 40.1 Surawicz CM (July 2008). "Role of probiotics in antibiotic-associated diarrhea, Clostridium difficile-associated diarrhea, and recurrent Clostridium difficile-associated diarrhea". J. Clin. Gastroenterol. 42 Suppl 2: S64–70. DOI:10.1097/MCG.0b013e3181646d09. PMID 18545161. Research Blogging.
- ↑ Schwan A, Sjölin S, Trottestam U, Aronsson B (October 1983). "Relapsing clostridium difficile enterocolitis cured by rectal infusion of homologous faeces". Lancet 2 (8354): 845. PMID 6137662. [e]
- ↑ McFarland LV (April 2006). "Meta-analysis of probiotics for the prevention of antibiotic associated diarrhea and the treatment of Clostridium difficile disease". Am. J. Gastroenterol. 101 (4): 812–22. DOI:10.1111/j.1572-0241.2006.00465.x. PMID 16635227. Research Blogging.
- ↑ Klarin B, Wullt M, Palmquist I, Molin G, Larsson A, Jeppsson B (September 2008). "Lactobacillus plantarum 299v reduces colonisation of Clostridium difficile in critically ill patients treated with antibiotics". Acta Anaesthesiol Scand 52 (8): 1096–102. DOI:10.1111/j.1399-6576.2008.01748.x. PMID 18840110. Research Blogging.
- ↑ Pépin J, Valiquette L, Alary ME, Villemure P, Pelletier A, Forget K et al. (2004). "Clostridium difficile-associated diarrhea in a region of Quebec from 1991 to 2003: a changing pattern of disease severity.". CMAJ 171 (5): 466-72. DOI:10.1503/cmaj.1041104. PMID 15337727. PMC PMC514643. Research Blogging.
- ↑ Barreto AR et al (2008). "Fucoidin Prevents Clostridium difficile Toxin-A-Induced Ileal Enteritis in Mice". Dig. Dis. Sci. 53(4): 990-6. PMID 17805968. [e]
- ↑ Gerber M, Ackermann G (2008). "OPT-80, a macrocyclic antimicrobial agent for the treatment of Clostridium difficile infections: a review". Expert. Opin. Investig. Drugs 17(4): 547–53. PMID 18363518. [e]
- ↑ Sorg JA, Sonenshein AL (2008). "Bile salts and glycine as cogerminants for Clostridium difficile spores". J Bacteriol 190(7): 2505-12. PMID 18245298. [e]