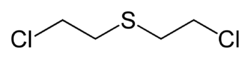
Mustard gas
Mustard gas refers to a family of potentially lethal but primarily casualty-producing chemical weapons introduced in the First World War (WW1) and subsequently improved in World War II (WWII). The first version was impure 2,2'-dichloroethyl sulfide, code-named "Yellow Cross" or "Lost" by the Germans and "H" in the modern U.S. system. Originally produced by the Levinstein reaction, it could be purified by distillation into the more potent "HD". During WWII, the U.S. developed a family of much more toxic nitrogen mustards gasses, called the "HN" series. After their declassification at the end of the war, they proved to be the basis of some of the first effective antineoplastic agents for cancer chemotherapy.
World War I
While it had been synthesized in 1860, mustard gas was first used in warfare in September 1917. German forces employed it against Russians at Riga. Considerably more toxic by weight than earlier chemical weapons, it became a regular part of bombardments until the end of the war.
As opposed to other agents in use at that time, it could penetrate unbroken skin. A mask alone was insufficient protection. Its onset of toxic effects were usually delayed, so there was a terrifying sense of uncertainty after being shelled.
World War II
Both sides stockpiled mustard but did not use it, with the possible exception of the Japanese against the China. There was, however, a major mustard gas contamination incident in Bari, Italy when German aircraft unknowingly bombed an ammunition ship carrying the theater reserve of the agent.
Germany conducted the Nazi mustard gas experiments on concentration camp prisoners.
Chemistry and biochemistry of mustard gasses
The chemistry and health effects of mustard gas have been thoroughly studied and comprehensive reviews are available.[1] Both mustard gas and the related amine gas weapons generally have a central Lewis base (electron donor), either an atom or a group, that can undergo nucleophilic attack of the terminal halogenated carbon(s) to form a cyclic cation (phosphonium, ammonium, or oxonium) with subsequent release of a halogen ion. The reactive phosphonium cation then reacts with water, opening the cyclic intermediate to form the hydroxide analog of the starting compound and releasing an acidic proton. Thus, the overall reaction releases hydrochloric acid (HCl), which causes blistering of any exposed tissues. The reactive cyclic phosphonium ion also serves as an efficient alkylating reagent and can therefore react with, and attach to, biological chemicals such as DNA and proteins. Even if one subjected to mustard gas survives the initial blistering tissue damamge, subsequent cancers arising from DNA damage (alkylation, cross-linking) often lead to later death.
Synthesis
The prototypical mustard gas, bis-(2-chloroethyl)sulfide, can be synthesized several different methods.The chemistry and health effects of mustard gas have been thoroughly studied and comprehensive reviews are available.[2]. Pure sulfur mustard can be prepared using Meyer's method of reacting thiodiglycol with phosphorus trichloride (PCl3) or other chlorinating agents including phosgene, thionyl chloride or concentrated hydrochloride. An alternative approach, which was used to produced World War I product, is to use the Levinstein method in which sulfur dichloride is reacted with ethylene to first produce 2-chloroethylsulfenyl chloride which is converted by a second addition of ethylene to mustard gas.
- Meyer Method 3 (HO-CH2=CH2)-S-(CH2=CH2-OH) + 2 PCl3 --> 3 S(CH2CH2Cl)2 + 2 P(OH)3
- Levinstein Method SCl2 + 2 H2C=CH2 --> (ClCH2CH2)2S
The Meyer method produces a purer product.
Biological chemistry
Mustard gas, at neutral pH, alkylates purines, pyrimidines, nucleosides, and nucleotides, preferentially at the N-7 atom of guanine and the N-1 atom of adenine. Reactions with the 0-6 atom and N-2 atom of guanine and N-6 atom of adenine have also been reported. Because mustard gas is bifunctional, that is to say it contains two reactive haloethylene groups, it is much more damaging than the monofunctional analog. This is because it can form either cyclic DNA adducts or can cross-link two separate DNA strands across a double helix, and such DNA damage is much harder to repair by the DNA repair and excision mechanisms present in biological entities. In addition, mustard gas can also attach to the proteins, such as DNA repair enzymes, RNA and phospholipids.
Nonproliferation
The Meyer process has advantages for nonproliferation, as some of its precursors are fairly specific to chemical weapons productions when shipped in large quantities. In contrast, the precursors of the Levinstein process are used in a wide range of industrial processes; the main control on this reaction is the more complex manufacturing requiring the manipulation of hot gases and liquids.
Thiodiglycol has relatively few other uses, and is a Scheduled substance under the Chemical Weapons Convention. By controlling the production and sales of thiodiglycol, there is some chance of controlling the production of mustard gas. While there are more applications for thionyl chloride, both are in the least restrictive Schedule 3 of the CWC.
Iraq, under Saddam Hussein, made huge purchases of thiodiglycol, whose other main application is in the formulation of ink for ball-point pens. While this could have been recognized and controlled, it was not.