Boiling point
The boiling point of a liquid—commonly "the temperature at which it boils", i.e., changes to gas—is the temperature at which the vapor pressure of the liquid equals the external environmental pressure surrounding the liquid. Since the boiling point of a liquid varies with surrounding environmental pressure, there is no single temperature at which a liquid boils. Instead there are an infinite number of boiling points corresponding to the infinite values of the surrounding pressure. The only single boiling point of note is the "normal boiling point" (as discussed below in more detail) which is the special case at which the boiling point occurs when the liquid's vapor pressure equals a surrounding environmental pressure of 1 atmosphere (101.325 kilopascals). That is the boiling point that most lay people incorrectly consider to be "the" boiling point when in fact a liquid has an infinite number of boiling points. At a given surrounding pressure, each liquid will have its own unique boiling point temperature.
The boiling point of a liquid may also be defined as the temperature at which it changes state from a liquid to a gas throughout the bulk of the liquid. At that point, bubbles of vapor which form below the surface of the liquid contain vapor at a pressure which matches the external pressure. Therefore, they are not crushed by the surrounding liquid and their buoyancy causes them to rise through to the surface of the liquid and give the familiar appearance of a boiling liquid.[1][2][3]
A liquid in a vacuum environment has a lower boiling point than when the liquid is at atmospheric pressure. A liquid in a high pressure environment has a higher boiling point than when the liquid is at atmospheric pressure. In other words, as noted above, the boiling point of a liquid varies with and depends upon the surrounding environmental pressure. The boiling point increases with increased pressure up to the critical point, where the gas and liquid properties become identical. The boiling point cannot be increased beyond the critical point. Likewise, the boiling point decreases with decreasing pressure until the triple point is reached. The boiling point cannot be reduced below the triple point.
Liquids may change to a vapor at temperatures below their boiling points through the process of evaporation. Evaporation is a surface phenomenon in which molecules located near the vapor/liquid surface escape into the vapor phase. On the other hand, boiling is a process in which molecules anywhere in the liquid escape, resulting in the formation of vapor bubbles within the liquid.
The normal boiling point
The normal boiling point (also called the atmospheric boiling point or the atmospheric pressure boiling point) of a liquid is the special case in which the vapor pressure of the liquid equals 101.325 kilopascals (kPa).[4][5]
Usually, boiling points are published with respect to the atmospheric pressure (101.325 kPa or 1 atm) at sea level, which is also known as the standard atmosphere.[6] In other words, published boiling points of liquids are usually the normal boiling points of the liquids.
Calculation of the normal boiling point
The heat of vaporization is the amount of heat required to convert or vaporize a saturated liquid (i.e., a liquid at its boiling point) into a vapor.
If the heat of vaporization and the vapor pressure of a liquid at a certain temperature is known, the normal boiling point can be calculated by using this form of the Clausius-Clapeyron equation:[7][8]
| where: | |
| = the normal boiling point, K | |
| = the molar gas constant, 8.314 J · K –1 · mol –1 | |
| = is the vapor pressure at a given temperature, kPa | |
| = atmospheric pressure, kPa | |
| = the heat of vaporization of the liquid, J · mol –1 | |
| = the given temperature, K | |
| = the natural logarithm to the base e |
Saturation temperature and pressure
A saturated liquid contains as much thermal energy (i.e., heat) as it can without boiling and, conversely, a saturated vapor contains as little thermal energy as it can without condensing.
The term saturation temperature means boiling point and it is the temperature for a corresponding saturation pressure at which a liquid boils into its vapor phase. The liquid can be said to be saturated with thermal energy. Any addition of thermal energy results in a phase change.
If the pressure in a system remains constant (i.e., the system is isobaric), a vapor at saturation temperature will begin to condense into its liquid phase as thermal energy is removed. Similarly, a liquid at saturation temperature and pressure will boil into its vapor phase as additional thermal energy is applied.
The saturation pressure is the pressure for a corresponding saturation temperature at which a liquid boils into its vapor phase. Saturation pressure and saturation temperature have a direct relationship: as saturation pressure is increased so is saturation temperature.
If the temperature in a system remains constant (i.e., the system is isothermal), vapor at saturation pressure and temperature will begin to condense into its liquid phase as the system pressure is increased. Similarly, a liquid at saturation pressure and temperature will tend to boil into its vapor phase as the system pressure is decreased.
Relation between the normal boiling point and the vapor pressure of liquids
At any given temperature, for a specific substance, there is a pressure at which the gas of that specific substance is in equilibrium with its liquid or solid forms — i.e., when the rates at which molecules escape from and return into the vaporizing liquid or solid are equal. This is the vapor pressure of the specific substance at that temperature.
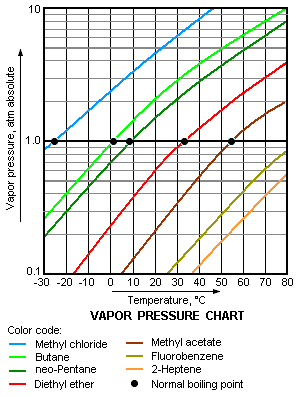
The vapor pressure chart to the right has graphs of the vapor pressures versus temperatures for a variety of liquids.[9] As can be seen in the chart, the liquids with the highest vapor pressures have the lowest normal boiling points.
For example, at any given temperature, methyl chloride has the highest vapor pressure of any of the liquids in the chart. It also has the lowest normal boiling point, which is where the vapor pressure curve of methyl chloride (the blue line) intersects the horizontal pressure line of one atmosphere (atm) of absolute vapor pressure.
In terms of intermolecular forces, the boiling point represents the point at which the liquid molecules possess enough thermal energy to overcome the various intermolecular attractions binding the molecules into the liquid. Therefore the boiling point is also an indicator of the strength of those attractive forces. The higher the intermolecular attractive forces are, the more difficult it is for molecules to escape from the liquid and hence the lower is the vapor pressure of the liquid. The lower the vapor pressure of the liquid, the higher the temperature must be to initiate boiling. Thus, the higher the intermolecular attractive forces are, the higher is the normal boiling point.[10]
Boiling point as a reference property of a pure compound
As can be seen from the above plot of the logarithm of the vapor pressure vs. the temperature for any given pure chemical compound, its normal boiling point can serve as an indication of that compound's overall volatility. A given pure compound has only one normal boiling point, if any, and a compound's normal boiling point and melting point can serve as characteristic physical properties for that compound, listed in reference books. The higher a compound's normal boiling point, the less volatile that compound is overall, and conversely, the lower a compound's normal boiling point, the more volatile that compound is overall. Some compounds decompose at higher temperatures before reaching their normal boiling point, or sometimes even their melting point. For a stable compound, the boiling point ranges from its triple point to its critical point, depending on the external pressure. Beyond its triple point, a compound's normal boiling point, if any, is higher than its melting point. Beyond the critical point, a compound's liquid and vapor phases merge together into one phase, which may be called a superheated gas. At any given temperature, if a compound's normal boiling point is lower, then that compound will generally exist as a gas at atmospheric external pressure. If the compound's normal boiling point is higher, then that compound can exist as a liquid or solid at that given temperature at atmospheric external pressure, and will so exist in equilibrium with its vapor (if volatile) if its vapors are contained. If a compound's vapors are not contained, then some volatile compounds can eventually evaporate away in spite of their higher boiling points.
In general, compounds with ionic bonds have high normal boiling points, if they do not decompose before reaching such high temperatures. Many metals have high boiling points, but not all. Very generally—with other factors being equal—in compounds with covalently-bonded molecules, as the size of the molecule (or molecular mass) increases, the normal boiling point increases. When the molecular size becomes that of a macromolecule, polymer, or otherwise very large, the compound often decomposes at high temperature before the boiling point is reached. Another factor that affects the normal boiling point of a compound is the polarity of its molecules. As the polarity of a compound's molecules increases, its normal boiling point increases, other factors being equal. Closely related is the ability of a molecule to form hydrogen bonds (in the liquid state), which makes it harder for molecules to leave the liquid state and thus increases the normal boiling point of the compound. Simple carboxylic acids dimerize by forming hydrogen bonds between molecules. A minor factor affecting boiling points is the shape of a molecule. Making the shape of a molecule more compact tends to lower the normal boiling point slightly compared to an equivalent molecule with more surface area.
Most volatile compounds (anywhere near ambient temperatures) go through an intermediate liquid phase while warming up from a solid phase to eventually transform to a vapor phase. By comparison to boiling, a sublimation is a physical transformation in which a solid turns directly into vapor, which happens in a few select cases such as with carbon dioxide at atmospheric pressure. For such compounds, a sublimation point is a temperature at which a solid turning directly into vapor has a vapor pressure equal to the external pressure.
Impurities and mixtures
In the preceding section, boiling points of pure compounds were covered. Vapor pressures and boiling points of substances can be affected by the presence of dissolved impurities (solutes) or other miscible compounds, the degree of effect depending on the concentration of the impurities or other compounds. The presence of non-volatile impurities such as salts or compounds of a volatility far lower than the main component compound decreases its mole fraction and the solution's volatility, and thus raises the normal boiling point in proportion to the concentration of the solutes. This effect is called boiling point elevation. As a common example, salt water boils at a higher temperature than pure water.
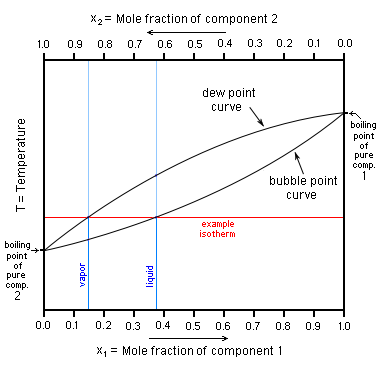
In other mixtures of miscible compounds (components), there may be two or more components of varying volatility, each having its own pure component boiling point at any given pressure. The presence of other volatile components in a mixture affects the vapor pressures and thus boiling points and dew points of all the components in the mixture. The dew point is a temperature at which a vapor condenses into a liquid. Furthermore, at any given temperature, the composition of the vapor is different from the composition of the liquid in most such cases. In order to illustrate these effects between the volatile components in a mixture, a boiling point diagram is commonly used. Distillation is a process of boiling and [usually] condensation which takes advantage of these differences in composition between liquid and vapor phases.
References
- ↑ H. Stephen Stoker (2006). General, Organic and Biological Chemistry, 4th Edition. Cengage Learning, page 165. ISBN 0-618-73063-X.
- ↑ Edward W. Washburn (1915). An Introduction to the Principles of Physical Chemistry, First Edition. McGraw-Hill, page 55. Library of Congress Control No. 15024872.
- ↑ Spencer L. Seager and Michael R. Slabaugh (2010). Chemistry for Today: General, Organic, and Biochemistry, 7th Edition. Brooks Cole, page 187. ISBN 0-538-73332-2.
- ↑ IUPAC Recommendations 1994 (page 537)
- ↑ David R. Lide (Editor) (2004). CRC Handbook of Chemistry and Physics, 85th Edition. CRC Press, page 3-1. ISBN 0-8493-0485-7.
- ↑ IUPAC Goldbook definition
- ↑ John M. Seddon and Julian D. Gale (2001). Thermodynamics and Statistical Mechanics. Royal Society of Chemistry. ISBN 0-85404-632-1.
- ↑ The Clausius-Clapeyron Equation (Professor Chung Chieh, University of Waterloo, Canada)
- ↑ Perry, R.H. and Green, D.W. (Editors) (1997). Perry's Chemical Engineers' Handbook, 7th Edition. McGraw-Hill. ISBN 0-07-049841-5.
- ↑ The Forces Between Molecules (University of Florida Chemistry Department website)
- Pages using ISBN magic links
- Editable Main Articles with Citable Versions
- CZ Live
- Engineering Workgroup
- Chemistry Workgroup
- Physics Workgroup
- Chemical Engineering Subgroup
- Articles written in American English
- Advanced Articles written in American English
- All Content
- Engineering Content
- Chemistry Content
- Physics Content
- Chemical Engineering tag

![{\displaystyle T_{B}={\Bigg (}{\frac {\,R\,[\,\ln(P_{0})-\ln(101.325)\,]}{\Delta H_{\mathrm {vap} }}}+{\frac {1}{T_{0}}}{\Bigg )}^{-1}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/045b915b89edf52e1f5e04a85fc8b4d3e58e4cdb)