Interspike interval histogram: Difference between revisions
imported>Gareth Leng |
imported>Gareth Leng |
||
| Line 19: | Line 19: | ||
==External links== | ==External links== | ||
Examples of recent papers | Examples of recent papers | ||
* | * Witham CL, Baker SN (2007)Network oscillations and intrinsic spiking rhythmicity do not covary in monkey sensorimotor areas ''J Physiol'' [http://jp.physoc.org/cgi/content/full/580/3/801 508:801-813] | ||
Witham CL, Baker SN ( | |||
Network oscillations and intrinsic spiking rhythmicity do not covary in monkey sensorimotor areas | |||
* [http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pubmed&pubmedid=16212763 Another recent full paper] | * [http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pubmed&pubmedid=16212763 Another recent full paper] | ||
Revision as of 11:09, 9 January 2008
An interspike-interval histogram is one of the ways by which experimental neurophysiologists analyse the electrical behaviour of neurons in the brain.
One of the most important ways by which neurons in the brain carry information is by means of electrical signals. At rest, neurons have a ‘resting membrane potential’ of typically about -70 mV: this is the potential difference between the inside of the cell and the external environment. However, this membrane potential is continually being disturbed by the chemical signals released by other neurons. In the brain a neuron might be receiving inputs from as many as ten thousand other neurons, these inputs usually consist of brief excitations (EPSPs; excitatory post-synaptic potentials) or of brief inhibitions (IPSPs; inhibitory post-synaptic potentials) that either make the membrane less negative (depolarisations) or more negative (hyperpolarisations). If a neuron becomes sufficiently depolarised, perhaps because of a flurry of EPSPs, then the membrane potential may reach the critical threshold for triggering an action potential (commonly called a ‘’spike’’). An action potential is a very large and rapid rise in the cell membrane potential, that lasts for only about one millisecond before the membrane potential returns again to around its resting potential. These spikes are propagated along the axons of a neuron, to reach the nerve terminals, where they can trigger the release of chemical messengers to affect other neurons.
Thus spikes are a very important way by which neurons in the brain carry information. The spike itself is an all-or-none phenomenon, so information is coded not in the amplitude of a spike but in the timing of spikes. Accordingly, electrophysiologists, who study the electrical behaviour of neurons, are interested in the patterning of spikes in particular neurons. The patterning of spike activity is influenced by three things: a) the intrinsic properties of the neuron, especially the properties of its membrane. The neurons in the brain are very diverse; there are very many different subpopulations of neurons that have quite different intrinsic properties b) network interactions, because spike activity in one neuron might have feedback effects on that neuron because of the changes that it produces in reciprocally connected neurons and c) the nature of the inputs to that neuron.
One of the simplest ways to study the patterning of spike activity in a neuron is to record the spikes from a single neuron, using a fine microelectrode, and then to construct an interspike interval histogram. This is simply a plot of the distribution of the observed times between spikes (t1, t2, t3, etc) collected in ‘bins’ of fixed width, say 1 ms. Typically, such a histogram would be constructed from about 2000 such intervals; if a neuron is firing at an average rate of about 5 spikes/s this will need a recording of 5-10 min of activity.
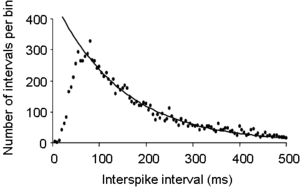
The figure on the right shows the interspike interval histogram recorded from a single neuron in the hypothalamus. Immediately after a spike, a neuron has an absolute refractory period in which it is unable to fire another spike, so the first few bins of the histogram will be empty – the histogram starts from zero. After this, the neuron has a relative refractory period, during which it is hard, but not impossible, to trigger another spike, and as this relative refractory period gradually decays, the interspike interval distribution rises to its peak value – the mode of the histogram, in this example at about 50 ms. The tail of the histogram (from about 50 ms on) declines exponentially to zero, as shown by the fitted line. This exponentially declining tail is an indication that, after about 50 ms after a spike, spikes are triggered by an essentially random process. This follows because the distribution of inter-event times for a wholly random process fits a single negative exponential, as first observed by Poisson.
In this example, we see that this particular neuron has quite a prolonged period of relative inexcitability after each spike, and might predict that this cell is one that shows a long hyperpolarsising after-potential (HAP) after each spike. For this particular cell, this is wholly correct; the cell is an oxytocin cell, and in these particular cells spikes trigger a large influx of calcium into the cell, and (in these particular cells) this subsequently causes a prolonged opening of a class of calcium-activated potassium channels, and this produces a relatively long HAP.
External links
Examples of recent papers
- Witham CL, Baker SN (2007)Network oscillations and intrinsic spiking rhythmicity do not covary in monkey sensorimotor areas J Physiol 508:801-813
- Another recent full paper