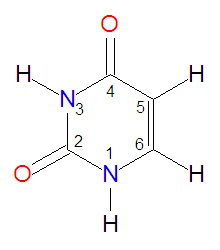
Uracil
|
| |||||||
| uracil | |||||||
| |||||||
| Uses: | RNA base | ||||||
| Properties: | |||||||
| Hazards: | |||||||
| |||||||
Uracil is one of the bases found in RNA. In RNA, the H1 proton is removed and the N1-nitrogen atom is bound the the C1' carbon of ribose. It is analogous in structure to thymidine, which can be thought of as 5-methyl-uracil, and it usually forms base pairs with adenine (A) in RNA. In an adenine-uracil base pair, the O4 oxygen atom of uracil forms a hydrogen bond with one of the amino protons (H61) of adenine. A second hydrogen bond is formed between the uracil H3 proton and the adenine N1 nitrogen atom. Like other nucleotide bases, it is subject to oxidative damage.
Substituted uracil in DNA
Oxidative damage to cytodine and thymine in DNA can result in the presence of substituted uracil bases in DNA, where they are normally not found. For example, the oxidative deamination of cytosine in DNA can lead to both 5-hydroxy-uracil (5-HOU) and 5,6-dihydroxy-uracil.[1],[2]
The guanine-cytosine to adenine-thymidine transition mutation is the most common base substitution mutation due to oxidative damage in DNA,[3] and the formation of 5-HOU is the largest contributor for this mutation, because the base that was formally a cytosine base paired with guanine, is now a uracil derivative that can form a base pair with adenine.[4],[5] Subsequent DNA replication can pair an adenine across from the 5-hydroxy-uracil rather than the correct guanine that was present in the parent DNA.
Oxidative damage to thymine, due to oxidation of the 5-methyl group, also produces substituted uracil bases in DNA.[6]Such oxidation produces 5-hydroxymethyluracil and 5-formyluracil. Quantum mechanics calculations show that 5-formyluracil can also base pair with adenine,[7] suggesting that this oxidatively damaged base could also cause DNA mutations.
References
- ↑ M. Dizdarouglu, E. Holwitt, M. P. Hagan & W. F. Blakely (1986). "Formation of cytosine glycol and 5,6-dihydroxycytosine in deoxyribonucleic acid on treatment with osmuim tetroxide". Biochem. J. 235: 531-536.
- ↑ D.A. Kreutzer and J. M. Essigman (1998). "Oxidized, deaminated cytosines are a source of C-> T transitions in vivo". Proc. Natl. Acad. Sci. USA 95: 3578-3582.
- ↑ R. M. Schaaper, B. N. Danforth and B.W. Glickman (1986). "Mechanisms of spontaneous mtagenisis: an analysis of the spectrum of spontaneous mutationin the Escherichia coli lacI gene". J. Mol. Biol. 189: 273-284.
- ↑ V. Thiviyanathan, A. Somasunderam, D. E. Volk, T. K. Hazra, S. Mitra and D. G. Gorenstein (2008). "Base-pairing properties of the oxidized cytosine derivative, 5-hydroxy uracil". Biophys. Biochem. Res. Commun. 366: 752-757.
- ↑ D. E. Volk, V. Thiviyanathan, A. Somasunderam and D. G. Gorenstein (2006). "Ab initio Base-pairings Energies of Uracil and 5-Hydroxy Uracil with Standard DNA Bases at the BSSE-free DFT and MP2 Theory Levels". Org. Biomol. Chem. 4: 1741-1745.
- ↑ E. Mullaat, P. H. M. Lohman, F. Brendes and J. Vijg (1990). "DNA damage metabolism and aging". Mutat. Res. 237: 189-210.
- ↑ D. E. Volk, V. Thiviyanathan, A. Somasunderam and D. G. Gorenstein (2007). "Ab initio base-pairing eneergies of an oxidized thymine product, 5-formyluracil, with standard DNA bases at the BSSE-free DFT and MP2 theory levels" 5: 1554-1558.