Petrochemicals
Petrochemicals are chemical products made from the hydrocarbons present in raw natural gas and petroleum crude oil. The largest petrochemical manufacturing industries are to be found in the United States of America, Western Europe, Asia and the Middle East.
A relatively small number of hydrocarbon feedstocks form the basis of the petrochemical industries, namely methane, ethylene, propylene, butanes, butadiene, benzene, toluene and xylenes.[1][2]
As of 2007, there were 2,980 operating petrochemical plants in 4,320 locations worldwide.[3] The petrochemical end products from those plants include plastics, soaps, detergents, solvents, paints, drugs, fertilizer, pesticides, explosives, synthetic textile fibers and rubbers, flooring and insulating materials and much more.
Petrochemicals are found in such common consumer products as aspirin, cars, clothing, compact discs, video tapes, electronic equipment, furniture, and a great many others.[4]
Feedstocks sources
The adjacent diagram schematically depicts the major hydrocarbon sources used in producing petrochemicals are:[1][2][5][6]
- Methane, ethane, propane and butanes: Obtained primarily from natural gas processing plants.
- Naphtha obtained from petroleum refineries.
- Benzene, toluene and xylenes, as a whole referred to as BTX and primarily obtained from petroleum refineries by extraction from the reformate produced in catalytic reformers.
- Gas oil obtained from petroleum refineries.
Methane and BTX are used directly as feedstocks for producing petrochemicals. However, the ethane, propane, butanes, naphtha and gas oil serve as optional feedstocks for steam-assisted thermal cracking plants referred to as steam crackers that produce these intermediate petrochemical feedstocks:
- Ethylene
- Propylene
- Butenes and butadiene
- Benzene
In 2007, the amounts of ethylene and propylene produced in steam crackers were about 115 Mt (megatonnes) and 70 Mt, respectively.[7] The output ethylene capacity of large steam crackers ranged up to as much as 1.0 – 1.5 Mt per year.[8][9]
Steam crackers are not to be confused with steam reforming plants used to produce hydrogen and ammonia.
Worldwide usage of optional steam cracking feedstock sources
As of 2004, the percentage of the worldwide steam cracking plants using each of the optional steam cracking feed sources was:[10]
- Ethane: 35%
- Propane: 9%
- Butanes: 3%
- Naphtha: 45%
- Gas oil: 5%
- Other: 3 %
The effect of feedstock on the steam cracking yields of intermediate petrochemical products
The effect of feedstock selection upon the yields of steam cracking products is summarized in the table below:
| ||||||||||||||||||||||||||||||||||||||||||||||||
Feedstocks and example petrochemical products
The table below includes some representative examples of the petrochemical end products produced from the eight hydrocarbon feedstocks – methane, ethylene, propylene, butenes, butadiene, benzene, toluene and xylenes:
| methane | ethylene | propylene | butenes and butadienes | benzene | toluene | xylenes |
|---|---|---|---|---|---|---|
| hydrogen | polyethylene | polypropylene | styrene-butadiene rubber (SBR) | styrene | benzoic acid | phthalic anhydride |
| ammonia | ethanol | isopropanol | methyl tert-butyl ether (MTBE) | polystyrene | toluene diisocyanate | polyesters |
| methanol | ethylene glycol | propylene glycol | polybutadiene | phenol | polyurethanes | dimethyl terephthalate |
| methyl chloride | vinyl acetate | allyl chloride | acrylonitrile-butadiene-styrene (ABS) | cumene | caprolactam | terephthalate acid |
| carbon black | perchloroethylene | acrylonitrile | polybutenes | aniline | nylons | polyethylene terephthalate |
| acetylene | polyvinyl acetate | acrylic acid | methyl ethyl ketone (MEK) | adipic acid | polyureas | dioctyl phthalate |
| formaldehyde | glycol ethers | epoxy resins | tert-butanol | nylons |
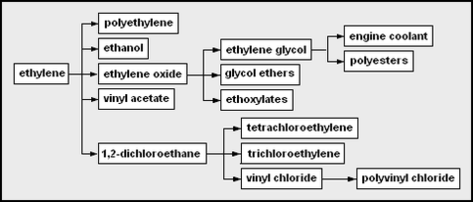
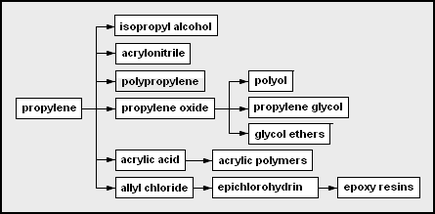
The chemicals that can be produced from each of the major petrochemical feedstocks can be depicted as "network diagrams". For example, these are the network diagrams for the chemicals that may be derived from ethylene and from propylene as the petrochemical feedstocks:
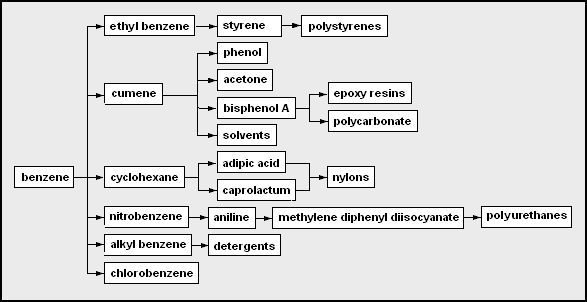
As another example, this is the network diagram for the chemicals that may be derived from benzene as the petrochemical feedstock:
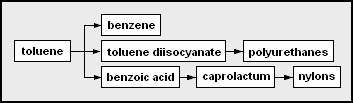
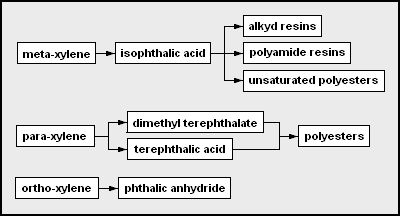
As more examples, these are the network diagrams for the chemicals that may be derived from toluene and from xylenes as the petrochemical feedstocks:
References
- ↑ 1.0 1.1 Sami Matar and Lewis F. Hatch (2001). Chemistry of Petrochemical Processes. Gulf Professional Publishing. ISBN 0-0-88415-315-0.
- ↑ 2.0 2.1 2.2 Staff (March 2001). "Petrochemical Processes 2001". Hydrocarbon Processing: pp. 71-246. ISSN 0887-0284.
- ↑ Petrochemical Industry – Worldwide
- ↑ Petrochemicals Chart From the website of the National Petrochemical & Refiners Association
- ↑ SBS Polymer Supply Outlook
- ↑ Jean-Pierre Favennec (Editor) (2001). Petroleum Refining: Refinery Operation and Management. Editions Technip. ISBN 2-7108-0801-3.
- ↑ Hassan E. Alfadala, G.V. Rex Reklaitis and Mahmoud M. El-Halwagi (Editors) (2009). Proceedings of the 1st Annual Gas Processing Symposium, Volume 1: January, 2009 - Qatar, 1st Edition. Elsevier Science, pp. 402-414. ISBN 0-444-53292-7.
- ↑ Crackers capacities From the website of the Association of Petrochemicals Producers in Europe (APPE)
- ↑ Steam Cracking: Ethylene Production (PDF page 3 of 12 pages)
- ↑ Richard Meyers (2003). The Basics of Chemistry. Greenwood Press. ISBN 0-313-31664-3.