Bacillus subtilis: Difference between revisions
imported>Annette Kosiorowski |
imported>Annette Kosiorowski |
||
| Line 75: | Line 75: | ||
[http://www.epa.gov/oppt/biotech/pubs/fra/fra009.htm ''Bacillus subtilis final risk assessment'' (1997, February). In Environmental Protection Agency Website. Retrieved March 26, 2008] | [http://www.epa.gov/oppt/biotech/pubs/fra/fra009.htm ''Bacillus subtilis final risk assessment'' (1997, February). In Environmental Protection Agency Website. Retrieved March 26, 2008] | ||
[http://www.nature.com/nature/journal/v390/n6657/full/390249a0.html Kunst, F., Ogasawara, N., Moszer, I. ''et al''. '"The complete genome sequence of the gram-positive bacterium Bacillus subtilis." ''Nature.'' 1997. Volume 390. | [http://www.nature.com/nature/journal/v390/n6657/full/390249a0.html Kunst, F., Ogasawara, N., Moszer, I. ''et al''. '"The complete genome sequence of the gram-positive bacterium Bacillus subtilis." ''Nature.'' 1997. Volume 390. pp. 249-256. ] | ||
[ http://dx.doi.org/10.1016/j.peptides.2007.06.014 Wang, C.L., Ng, T.B., ''et al.'' "Induction of apoptosis in human leukemia K562 cells by cyclic lipopeptide from Bacillus subtilis natto T-2." ''Peptides.'' 2007. Volume 28, Issue 7. pp. 1344-1350.] | |||
Devine, Kevin M. (2000). "Bacillus subtilis: Genetics." In ''Encyclopedia of Microbiology, Second Edition''. Lederberg, Joshua (Ed.), New York: Academic Press. | Devine, Kevin M. (2000). "Bacillus subtilis: Genetics." In ''Encyclopedia of Microbiology, Second Edition''. Lederberg, Joshua (Ed.), New York: Academic Press. | ||
Lonenshein, A.L., Hoch, J.A., & Losick, R. (1993). ''Bacillus subtilis and other gram positive bacteria.'' Washington, D.C.: American Society for Microbiology. | Lonenshein, A.L., Hoch, J.A., & Losick, R. (1993). ''Bacillus subtilis and other gram positive bacteria.'' Washington, D.C.: American Society for Microbiology. | ||
Revision as of 22:46, 1 April 2008
Articles that lack this notice, including many Eduzendium ones, welcome your collaboration! |
Classification
Higher order taxa
Domain:Eubacteria
Phylum: Firmicutes
Class: Bacilli
Order: Bacillales
Family: Bacillaceae
Species
Genus: Bacillus
Species: subtilis
Description and significance
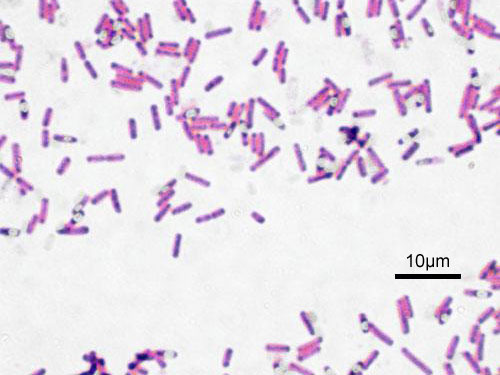
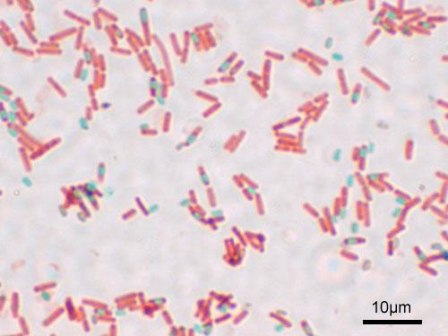
Bacillus subtilis is a gram-positive, rod-shaped, endospore-forming bacteria. It is regarded as an aerobe; however, it is also capable of growing and sporulating under anaerobic conditions when necessary. B. subtilis resides predominantly in soil, including low-nutrient soil. Due to its association with soil particles, it is also inevitably transferred to plants, foods, animals and even marine and freshwater habitats.
B. subtilis was one of the first bacteria studied by scientists. It was originally named Vibrio subtilis in 1835 by Christian Gottfried Ehrenberg and later renamed Bacillus subtilis by Ferdinand Cohn in 1872.
Currently, B. subtilis is a significant microorganism in the scientific research field, as well as in biotechnology and industry. Scientists often utilize B. subtilis as a model organism. B. subtilis becomes naturally competent during its transition between exponential growth and the stationary phase of growth, meaning it is able to bend and internalize DNA from a medium. Thus, it is easily manipulated genetically and a good laboratory microorganism. Due to its endospore-forming mechanisms, it is an especially excellent model system for cell differentiation. Furthermore, B. subtilis produces a variety of enzymes used in biotechnology and industry, including antibiotic production. Its status as a "generally regarded as safe" (GRAS) organism makes it an excellent industrial microorganism.
Genome structure
The genome sequence of Bacillus subtilis was completed and published in November 1997. B. subtilis contains a single, circular chromosome. It consists of 4,214,810 base pairs. The average G+C content is 43.5%, although ten regions have a G+C content significantly lower than average. There is a significant G-T skew at the third codon positions in common with other bacteria. Its leading strand has a 9% excess of G and 4% excess of T, as compared to the lagging strand. Approximately 87% of the genome consists of protein-coding regions. More than 74% of the all open reading frames and 94% of ribosomal genes are transcribed co-directionally with replication. Only 53% of the genes are represented once. The remaining belong to multigene families, which range in size from 2 to 77 gene copies. Approximately 220 transcriptional regulators have been identified in the genome. At the present time, the functions of only approximately 58% of the genes are known, while the functions of 42% are still unknown and being studied. Twelve percent of these genes with unknown functions have homologs in other organisms.
Replication of the B. subtilis genome proceeds similarly to most bacteria, with the splitting of the chromosome into two replication forks. B. subtilis has an origin of replication (oriC) and a terminus of replication (terC) located almost perfectly opposite each other on the genome. However, some studies have shown that the clockwise and counterclockwise replication forks actually differ in length by about 170 base pairs. Thus, once the clockwise replication fork reaches the terminus, its synthesis is terminated, while synthesis of the counterclockwise fork continues until it finally reaches the terminus.
Cell structure and metabolism
Bacillus subtilis is a prokaryotic cell, lacking membrane-bound organelles. It is enclosed by a cell wall, consisting of large amounts of peptidoglycan (gram-positive). It is rod, or bacillus, shaped and usually found in colonies with other B. subtilis. B. subtilis is flagellated and thus motile.
Cellular respiration in B. subtilis is mainly aerobic. Glycolysis and the Tricarboxylic Acid (TCA) cycle are complete and functional in the bacteria. However, it is able to grow under anaerobic conditions in the presence of nitrate, though growth is much slower.
B. subtilis has the ability to produce acid from a variety of sugars. In response to nutrient limitation, it also produces several enzymes (including proteases, amylases, cellulases, and lipases) and antibiotics. These enzymes are believed to help search for and degrade macromolecules to be used as energy sources, while the antibiotics are used to limit bacterial competition for the discovered energy sources. Furthermore, B. subtiliscontains 34 different two-component systems. Two-component systems consists of proteins that can detect environmental conditions and initiate the appropriate responses to these conditions. This large number of two-component systems suggests that B. subtilis can respond to a variety of environmental conditions effectively with a wide range of responses.
B. subtilis often exists as an endospore. As an endospore, B. subtilis becomes resistant to heat, acid, salt, and other harsh environmental conditions. Endospore formation is usually triggered by a lack of carbon, nitrogen or phosphate in the environment. This environmental "signal" is sensed by B. subtilis' two-component systems, which initiate a phosphorelay by transferring a phosphate to Spo0A, one of the early transcriptional regulators involved in endospore formation. Hundreds of genes in the B. subtilis genome are involved in this phosphorelay. Asymmetric cell division proceeds, producing a large mother cell and a smaller, ellipse-shaped spore. The mother cell first engulfs the spore, and, following a series of morphological changes, the spore cortex and spore coat are then formed. The mother cell lyses and releases the newly formed endospore, which can survive through the poor environmental conditions.
Ecology
B. subtilis is an important soil microorganism. It produces a variety of enzymes, which allow it to degrade many natural substrates and thus cycle nutrients in the soil. It is also capable of increasing nutrient availability to plants, stimulating plant growth, and suppressing plant diseases, increasing the overal performance of crops and other plants. Moreover, B. subtilis secretes antifungal antibiotics, which can control fungal diseases in plants and crops.
Pathology
B. subtilis is not pathogenic to humans, animals, or plants. Although it may contaminate certain foods, such as cocoa and spices, it rarely causes food poisoning. B. subtilis does produce one extracellular toxin, the enzyme subtilisin. Subtilisin can result in allergic reactions in some individuals; however, the allergic reaction will only occur following prolonged and repeated exposure.
Application to Biotechnology
B. subtilis is widely used in the field of biotechnology due to its ease at being manipulated and its generally low risk. B. subtilis produces a variety of enzymes useful to industry. The amylases it produces are used in the desizing of textiles and in starch modifications used to resize paper. The proteases it produces are used as additives in laundry detergents and products used for processing leather. B. subtilis also produces a wide array of antibiotics, including subtilin, surfactin, bacillomycin, bacilysin, and fengycin, which are used an antibacterial and antifungal medicines. The antifungal activity of B. subtilis straing QST713 is used as a biological control agent of fungal plant pathogens. B. subtilis is also used as a soil inoculant in horticulture and agriculture. Furthermore, B. subtilis has been used to convert explosives into harmless compounds and to safely discard radionuclide wastes. B. subtilis was also used as a biowarfare simulant in Project SHAD. Project SHAD was a series of chemical and biological warfare tests conducted by the U.S. military during the Cold War era. Military personnel were exposed to biological and chemical agents, including B. subtilis, to determine military vulnerabilities to such biowarfare agents and to determine appropriate response procedures.
B. subtilis is also used directly in the food industry. A strain of B. subtilis is used in the production of natto, a Japanese delicacy, and cheonggukjang, a similar Korean delicacy.
Current Research
"Induction of apoptosis in human leukemia K562 cells by cyclic lipopeptide from Bacillus subtilis natto T-2." (2007)
Recent cancer research has focused much attention on inducing apoptosis of cancerous cells, as well as finding natural products with cancer-fighting effects. This research study examined the potential anti-cancer effects of a cyclic lipopeptide isolated from the natto strain of Bacillus subtilis. This natto strain is safe to humans and can be found in a traditional Japanese food called "natto". The cyclic lipopeptide, in previous studies, exhibited anti-tumor properties. This study aimed to evaluate the cell growth-inhibiting and apoptosis-inducing effects of the cyclic lipopeptide from B. subtilis on human leukemia K562 cells. Researchers isolated the natto strain of B. subtilis directly from the Japanese food natto and obtained from it the cyclic lipopeptide. Leukemia K562 cells were treated with various concentrations of the cyclic lipopeptide, and fluorescent staining, flow cytometric analysis and Western blotting were used for analysis. Fluorescent staining showed that the cyclic lipopeptide did induce apoptosis in the leukemia cells. Flow cytometric analysis indicated that the number of apoptotic cells increased with increasing concentrations of cyclic lipopeptide. Western blotting revealed that the apoptosis could be associated with the proteins caspase-3 and poly(ADP-ribose)polymerase. Therefore, this study provided evidence that lipopeptides from the natto strain of B. subtilis do have anti-tumor activity and can inhibit the proliferation of leukemia cells by inducing their apoptosis. With further research, this B. subtilis strain may prove to be an effective therapeutic agent against cancer.
References
Bacillus Subtilis (2008, March 26). In Wikipedia, The Free Encyclopedia. Retrieved March 26, 2008
[ http://dx.doi.org/10.1016/j.peptides.2007.06.014 Wang, C.L., Ng, T.B., et al. "Induction of apoptosis in human leukemia K562 cells by cyclic lipopeptide from Bacillus subtilis natto T-2." Peptides. 2007. Volume 28, Issue 7. pp. 1344-1350.]
Devine, Kevin M. (2000). "Bacillus subtilis: Genetics." In Encyclopedia of Microbiology, Second Edition. Lederberg, Joshua (Ed.), New York: Academic Press.
Lonenshein, A.L., Hoch, J.A., & Losick, R. (1993). Bacillus subtilis and other gram positive bacteria. Washington, D.C.: American Society for Microbiology.