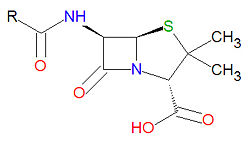
Penicillin
Penicillin refers to a class of antibiotic drugs that have a common -lactam core structure. The discovery of penicillin and development of its use in bacterial infections is one of the landmark scientific accomplishments of the 20th century.
Penicillin was discovered in 1928 by Scottish scientist Alexander Fleming as a crude extract of P. rubens. Fleming's student Cecil George Paine was the first to successfully use penicillin to treat eye infection (neonatal conjunctivitis) in 1930. The purified compound (penicillin F) was isolated in 1940 by a research team led by Howard Florey and Ernst Boris Chain at the University of Oxford. Fleming first used the purified penicillin to treat streptococcal meningitis in 1942. The 1945 Nobel Prize in Physiology or Medicine was shared by Chain, Fleming, and Florey for their work on penicillin.
Natural and semisythetic penicillins are produced by fungi: the original strain producing penicillin G (benzylpenicillin) was Penicillium notatum, although the industry changed to a higher-yielding P. chrysogenum.
Semisynthetic penicillins have precursor chemicals added to the culture media, which are incorporated into the antibiotic molecule. The modifications variously improve coverage of Gram-negative organisms, beta-lactamase resistance, or both. Another approach to resisting penicillinase is to co-administer a competive inhibitor of the destructive enzyme, such as clavulanic acid with amoxicillin.
For more information about specific penicillin drugs, see these pages: