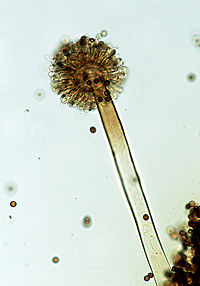
Aspergillus niger
| Aspergillus niger | ||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
 | ||||||||||||||||||||||
| Scientific classification | ||||||||||||||||||||||
| ||||||||||||||||||||||
| Binomial name | ||||||||||||||||||||||
| Aspergillus niger [1] |
Description and significance
Aspergillus niger is a filamentous fungus that has many applications in biotechnology.[1] This eukaryotic organism belongs to the Fungi kingdom and the Aspergillus genus. The use of microorganisms in biotechnology is common, however A. niger is considered to be one of the most essential of those microorganisms.[2]
The significance of A. niger is the industrial role that it plays in the production of proteins, enzymes and fermentation. It is capable of producing heterologous proteins, such as human cytokine interleukin -6. [3] This very useful microbe is even referred to as an "industrial workhorse" because of the frequent use in many applications.[4] [5]
Genome structure
The genome sequencing of Aspergillus niger can be performed with many advanced biological analysis techniques. Continued research in this microbe could lead to greater possibilities for the use of A. niger; as there are 7,500 genes that are not yet known.[5]
A. niger CBS 513.88 strain is the early ancestor of enzyme production strains that are currently used. CBS 513.88 strain was sequenced by large team that used a process called BAC walking, which required the use of an ordered set of large E. coli bacterial artificial chromosomes (BACs). The results from the study were published in January 2007 in Nature Biotechnology. Results indicated that the size of the genome of A. niger CBS 513.88 is 33.9-megabased with a total of 14,165 protein-coding genes. The assembled genome sequence expresses 468 DNA contigs, a set of overlapping DNA segments that stretch a total of 33.9 million unique bp (Mbp) that are arranged in 19 supercontigs. The average gene length is 1,572 bp and the number of tRNA genes is 269.[5]
Cell structure and metabolism
A. niger is a fungus, but it is specified as a mold. [6] It is an ascomycetous fungus that produces microscopic spores inside sacs or elongated cells called asci. A. niger is known to reproduce through asexual spores only, no sexual reproduction. As this microbe is an important host for production of various proteins and metabolites; it would be beneficial for strain improvement if there was an available sexual reproduction cycle.
The possibility of sexual reproduction allows for recombination of genes to have the opportunity to repair naturally occurring mutations and results in new genotypes and phenotypes for natural selection. Asexual reproduction has beneficial traits as well, it allows for dispersion of many genetically identical spores that do not require much metabolism in comparison to sexual reproduction.[7]
In 2007, the genome of strain CBS 513.88 was analyzed by Pel HJ et al, and genes that are associated with sexual reproduction were discovered.[5] However, those sexual reproduction related genes are not yet determined if deemed functional in fungi that is known to carry out only asexual reproduction.[6]
A. niger has a cell wall that is made up of chitin, 1,3- -glucan, 1,6- -glucan, 1,3- -glucan, galactosaminogalactan and galactomannan and the fungal cell wall determines the morphological features of the microbe.[5] Genes that are needed to produce those cell wall constituents were identified in the genome of CBS 513.88 strain. (Pel et al, 2007).
In order for this industrial workhorse to carry out its important tasks; the metabolism of A. niger consists of a “reaction network that comprises central carbon metabolism, catabolic pathways for 115 different carbon sources and 23 different nitrogen sources and anabolic pathways for the components of the biomass” (Andersen 'et al, 2008).
Ecology
A. niger grows aerobically on organic matter, therefore it can be found almost everywhere in environments that contain soil. Also, it is found in waste, decaying plant material and compost in outdoor environments. Moist indoor environments create a good habitat for the growth of mold. Such as in common household areas, on walls in bathrooms or other areas that have mositure. There are other indoor molds that also appear as black spores, which have similar resemblance to A. niger.[8]
Aspergillus niger has been exhibited to sustain growth in freezing temperatures, which indicates it as a thermotolerant that can also survive at very high temperatures. It’s thermotolerant abilities that enable growth in a wide temperature range from 6 to 47°C with a preferred optimum temperature at 35-37°C. The fungus is capable of growing over a very wide pH range, from 1.4 to 9.8 pH. The growth ability in various temperature ranges, pH ranges as well as the abundant amount of conidiospores allow the species to be continuously widespread. Conidiospores are distributed by air.[2]
Plant pathology
In plants, the Black Rot disease affects onions when black-colored spores of A. niger are present, appearance of black mold of the onions. This filamentous fungus causes diseases in peanuts and in grapes as well. [9]
Human pathology
A. niger has been linked to cases of human pulmonary infections. [10] The patient had cavitary lung disease and hemoptysis (coughing up blood) that developed foreign bodies in the respiratory tract with intractable metabolic acidosis that resulted in death.
Species belonging to the Aspergillus genus share similar traits of branching septate hyphal mold that can cause different diseases in hosts that either naturally have a weak immune system or weakened immune system from an unrelated disease. Aspergilloma is the mycelial growth of the fungus in a cavitary space. Of the Aspergillus species that cause disease, the highest number of cases from A. fumigatus (85%), A. flavus (10%) and A. niger (2%).[10]
Aspergillus species, including A. niger, releases oxalic acid (C2O4H2) which is a side product of the tricarboxylic acid cycle and is released as a mycotoxin. Mycotoxins are toxic secondary metabolites produced normally by Fungi. Oxalic acid complexes that have free calcium ions in blood and tissue form calcium oxalate. It is deposited mainly as monoclinic calcium oxalate monohydrate crystals when the concentration of the product has reached equilibrium. [11]
Uncommon infections caused by A. niger can be found in areas such as wounds, burns and lesions of mucus membranes and the alternative method of transmission of A. niger infection in healthy humans are through respiratory and digestive tracts.
Spores of A. niger that cause mycosis in the human ear is a common occurring health issue in tropical regions. From studies by Ibekwe and Okafor in 1983, it was determined that the fungus is a secondary invader, and not the causative organism. Based on the fact that the infected patients were treated with antibiotics before the isolation of A. niger from the ears. [12] Otomycosis, is the name of these ear infections that cause inflammation and mycelial growth on earwax on the skin of the external ear canal. Otomycosis is not considered a very serious condition as treatment options require topical anti-fungal medication to the infected ear.[2]
Animal pathology
Aspergillosis disease has been observed and documented in both humans and animals since it was first described in 1856. Depending on the immunologic state of the host and the species of Aspergillus involved, the seriousness of the disease varies. Aspergillus pathogens can affect potential hosts by acting as allergens or colonizers while trying to invade the host. Aspergillus is considered an opportunistic pathogen that targets mainly the respiratory tract and the brain to cause (systemic disease/secondary Aspergillosis).[13]
Tunve et al, 1999 case report of a horse with cerebral infarction caused by A. niger where findings concluded from microscopic examination of the cerebral lesion revealed many areas infiltrated by large numbers of “pale basophilic septate fungal hyphae” that resembled the morphology of A. niger hyphae. Necrosis, premature death of cells and living tissue affected many other areas of the animal’s body, such as spreading to the colon and mucous membranes resulting in hemorrhage.[13] It seems that Aspergillosis disease in animals and humans have similar symptoms.
A. niger in Biotechnology
In the field of biotechnology, A. niger is a valuable asset to Microbiology as it is an important microorganism because it has the ability to produce a variety of useful substances. It is therefore cultured for industrial production, often commercially for uses in making proteins, in the food industries and in pharmaceuticals.
Many enzymes, ie. citric acid, amylases, lipases, cellulose, xylanases, glucoamylase and proteases are produced by this fungus. A. niger is usually fermented to production those various enzymes. Using a solid state fermentation (SSF) method, glucoamylase was produced by A. niger where the fungus was inoculated with nutrient supplies and the study showed continuous production of the enzyme.[14] A. niger strain ATCC 1015 is most well-known as the strain that produces citric acid. Citric acid serves the purpose of improving taste, nutrition and shelf-life of food products in the food industry. A. niger is also referred to as a “versatile cell factory”[5].
A. niger can produce alpha-galacatosidase, which is an enzyme that is capable of breaking down certain non-digestible oligosaccharides in the digestive tract. Those non-absorbable oligosaccharides that are not broken down usually cause flatulence (gas). A. niger is used in a dietary supplement, Beano:[2] which can lower frequency of flatulence or other abdominal issues caused by those oligosaccharides that have not been fully digested.[15]
Current Research
"Effect of Ozone on Aspergillus niger Causing Black Rot Disease in Onion"
In vegetation, the Black Rot disease affects the onion, which is caused by Aspergillus niger. A. niger is a growing problem in the storage and transit of onions because an infected product would have growth of a black powdery spore masses of A. niger on the outer skin of the onion. For raw agricultural crops, ozone gas or ozone is added to the water for cleaning the crops. Usage of ozone as a disinfectant has shown to be effective in destroying pathogens and organisms that cause food to spoil; therefore it would be beneficial in extending storage life of foods. Ozone was approved by the FDA in 2002 for use in the food industry as it was titled with GRAS, “Generally Recognized as Safe” status in the United States. Samples of isolated A. niger infected onion samples were used and the isolate was placed to be grown in vitro on potato dextrose agar medium at 25 degrees C, for five days. Collection of A. niger spore was carried out through filtration into a suspension, and then transferred to tubes which were stored at 20 C to prevent premature spore germination. Spore suspension samples were put under different growth environments. Results on the effect of ozone on A. niger were that the ozone did not prevent spore germination of A. niger. On the contrary, ozone caused an increase in spore germination which was determined by the lower number of germinated spore in the control.
In another experiment, the inhibitory effects of ozone on the mycelium of Aspergillus niger was conducted (Chynoweth et al., 1997) [16] and the result was that at ozone concentration of 2.2mg/L, prevention of mycelium growth was successful. Comparison of these two experiments, one of ozone effect on mycelium of A. niger and the other on ozone effect of conidia of A. niger indicates that fungicidal effect of ozone did not affect the processes that cause spore germination. Spore germination in A. niger was not affected by ozone, except changes in colony morphology occurred. Spores treated with ozone and later cultured in vitro on PDA plate, caused change in sporulation; resulted in non-sporulating colonies that could be identified as grey patches in comparison to the surrounding sporulating colonies which were black in color. But, not all spores that were exposed to ozone resulted in unsuccessful sporulation, only spore suspensions that faced increased exposure of concentration to ozone were unsuccessful in sporulation. Resistant fungal spore can be effected by a pre-treatment of ozone to increase germination, and then with a higher mycelial inhibitory dose of ozone during onion storage. Ozone treatments produced many colonies with sterile mycelium in the next generation which can be controlled efficiently by adding the effective inhibitory ozone dose instead of that of the spore. From these studies, methods were conducted to help management of the black rot disease of the onion by A.niger during storage.[9]
"Regulation of Alternative Oxidase at the Transcription Stage in Aspergillus niger Under the Conditions of Citric Acid Production
Strain WU-2223L of A. niger, is one that produces citric acid and this strain has a cyanide-insensitive respiratory pathway that is catalyzed by alternative oxidase. Alternative oxidase can be found in many higher plant species, algae and fungi and its regulation under the conditions of citric acid production was deciphered from the transcription level of alternative oxidase gene (aoxl). Analyses of WU-2223L concluded there is only one copy of the aoxl on the chromosome. Alternative oxidase activity encoded by the aoxl gene is regulated at the transcription stage following the conditions that WU-2223L was tested under as well as for production of citric acid. This was determined by measuring alternative oxidase activities and transcription levels of aoxl gene.
An extra accumulation of citric acid by A. niger can be reasoned that the change in metabolism related to functioning of the cyanide=insensitive respiratory pathway catalyzed by alternative oxidase caused the extra citric acid production. Authors of this study hypothesized that the function of the alternative oxidase is essential to the increased production of citric acid by A. niger and that it must be important for the reoxidation of a large amount of NADH that is produced from the process of glycolysis. Results of this study concluded that alternative oxidase activity during the production of citric acid is regulated at the transcription stage. Therefore, A. niger WU-2223L has only one aoxl gene that encodes for an alternative oxidase on its homolog chromosome and to confirm that the contribution of the alternative oxidase is encoded by aoxl gene to production of citric acid can be done so by disrupting the aoxl gene causing an over expression of aoxl, which would result in overproduction of citric acid. [17]
"Recombinant bacterial hemoglobin alters metabolism of Aspergillus niger"
Aspergillus niger is used widely to produce enzymes and organic acids, however there is a drawback to the production when the ongoing issue of an limited supply of oxygen that is required in order for the respiratory metabolism of A. niger in industrial fermentation. The study was focused on the effect on the metabolism of A. niger when VHB is expressed in the cell; to further determine if this method of metabolic engineering could be a resolution to the problem that the fungus has with limited oxygen supply. Results from the study indicated that VHB did have a profound impact on the metabolism of A. niger under conditions where oxygen is limited. When VHB is present, there is less stress on the cells that usually are stressed during short supply of oxygen. Although the process of how VHB is able to fix the problem of limited oxygen are not concise yet, but based on the results obtained from the study, it is evident that VHB interferes with the redox metabolism of the cell and helps resolve the issue. [18]
References
- ↑ Oliveira JM, van der Veen D, De Graaff LH, Qin L. (2008). Efficient Cloning System for Construction of Gene Silencing Vectors in Aspergillus Niger. Applied microbiology and biotechnology. Vol. 80, Issue 5. pp 917-924. DOI 10.1007/s00253-008-1640-x
- ↑ 2.0 2.1 2.2 Schuster E, Dunn-Coleman N, Frisvad J, Van Dijck P. (2002). On the safety of Aspergillus niger – a review. Applied Microbiology and Biotechnology. Vol. 59, Numbers 4-5, pp 426-435. DOI 10.1007/s00253-002-1032-6
- ↑ Semova N, Storms R, John T, Gaudet P, Ulycznyi P, et al. (2006). Generation, annotation, and analysis of an extensive Aspergillus niger EST collection. BMC Microbiology. v.6:7. DOI 10.1186/1471-2180-6-7
- ↑ Andersen MR, Nielsen ML, Nielsen J. (2008). Metabolic model integration of the bibliome, genome, metabolome and reactome of Aspergillus niger. Molecular Systems Biology] 4: 178, pp 1-13. DOI 10.1038/msb.2008.12
- ↑ 5.0 5.1 5.2 5.3 5.4 5.5 Pel HJ, de Winde JH, Archer DB, Dyer PS, Hofmann G, et al. (2007). Genome sequencing and analysis of the versatile cell factory Aspergillus niger CBS 513.88. Nature Biotechnology, 25. pp 221-231. DOI 10.1038/nbt1282
- ↑ 6.0 6.1 Wadman M, de Vries RP, Kalkhove SIC, Veldink GA, Vliegenthart JFG. (2009). Characterization of oxylipins and dioxygenase genes in the asexual fungus Aspergillus niger. BMC Microbiology. v.9:59. DOI 10.1186/1471-2180-9-59
- ↑ Taylor JW, Jacobson DJ, Fisher MC. (1999). The evolution of asexual fungi: reproduction, speciation and classification. Ann Rev Phytopathology. Volume 37. pp 197–246. DOI 10.1146/annurev.phyto.37.1.197.
- ↑ Deepake, U., (2009) Aero-microbiological studies of Moisture Affected Buildings in the Indoor Environment. Journal of Young Investigators. Vol. 19, Issue 11.
- ↑ 9.0 9.1 9.2 Vijayanandraj, V.R., Nagendra Prasad, D., Mohan, N., Gunasekaran, M. (2006). Effect of Ozone on Aspergillus niger Causing Black Rot Disease in Onion. Ozone: Science & Engineering. Vol. 28, Issue 5. pages 347-350.
- ↑ 10.0 10.1 Roehrl MH, Croft WJ, Liao Q, Wang JY, Kradin RL. (2007). Hemorrhagic pulmonary oxalosis secondary to a noninvasive Aspergillus niger fungus ball. Virchows Archiv. Vol. 451, Number 6., pp 1067-1073. DOI 10.1007/s00428-007-0487-3 In a recent case report by Roehrl et al, 2007 conducted a study and had findings that detail a medical case of hemorrhagic pulmonary oxalosis secondary to a noninvasive Aspergillus niger fungus ball.
- ↑ Hope WW, Walsh TJ, Denning DW. (2005). The invasive and saprophytic syndromes due to Aspergillus spp. Med Mycol 43(Suppl 1):S207–S238 The crystal deposition of oxalate has toxic abilities that cause hemorrhage and tissue necrosis, which is premature death of tissue cells.
- ↑ Ibekwe, A.C., Okafor, J.I. (1983). Pathogenic organisms in chronic suppurative otitis media in Enugu, Nigeria. Trop Geogr Med 35:389-391.
- ↑ 13.0 13.1 Tunev SS, Ehrhart EJ, Jensen HE, Foreman JH, Richter RA, Messick JB. (1999). Necrotizing mycotic vasculitis with cerebral infarction caused by Aspergillus niger in a horse with acute typholocolitis. Vet Pathology. 1999 Jul:36(4):347-51.
- ↑ Varzakas TH, Roussos S, Arvanitoyannis IS. (2007. Glucoamylases production of Aspergillus niger' in solid state fermentation using a continuous counter-current reactor. International Journal of Food Science and Technology. Vol. 43, Issue 7. pp 1159-1168.
- ↑ Rajoka MI, Awan MS, Saleem M, Ayub N. (2008) Solid-state fermentation-supported enhanced production of α-galactosidase by a deoxyglucose-resistant mutant of Aspergillus niger and thermostabilization of the production process World Journal of Microbiology and Biotechnology. Vol. 25, Issue 2. pp 171-8. DOI 10.1007/s11274-008-9886-0
- ↑ Chynoweth, D., (1997). ‘‘Influence of Ozone on Growth and Accumulation of Microorganisms in Air Conditioning Systems’’, Proposal for The Canadian Institute for Advanced Research (CIAR), Canad.]
- ↑ 17.0 17.1 Hattori T, Kino K, Kirimura K. (2009). Regulation of Alternative Oxidase at the Transcription Stage in Aspergillus niger Under the Conditions of Citric Acid Production. Vol. 58, Issue 58. pp 321-325.
- ↑ 18.0 18.1 Hofmanna G, Dianoa A, Nielsen J. (2009). Recombinant bacterial hemoglobin alters metabolism of Aspergillus niger. Metabolic Engineering. Vol. 11, Issue 1. pp 8-12. DOI 10.1016/j.ymben.2008.07.002