Penicillin V: Difference between revisions
imported>David E. Volk mNo edit summary |
imported>David E. Volk (drug interactions) |
||
| Line 9: | Line 9: | ||
== Chemistry == | == Chemistry == | ||
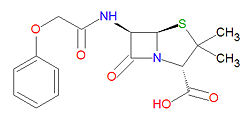
Penicillin V is stable against degradation by beta-[[lactamase]]s, including [[penicillinase]]s, and [[cephalosporinase]]s. Its IUPAC chemical name is (2S,5R,6R)-3,3-dimethyl-7-oxo-6-[[2-(phenoxy)acetyl]amino]-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylic acid and it has molecular formula C16H18N2O5S (MW = 350.3895 g/mol). | Penicillin V is stable against degradation by beta-[[lactamase]]s, including [[penicillinase]]s, and [[cephalosporinase]]s. Its IUPAC chemical name is (2S,5R,6R)-3,3-dimethyl-7-oxo-6-[[2-(phenoxy)acetyl]amino]-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylic acid and it has molecular formula C16H18N2O5S (MW = 350.3895 g/mol). | ||
== Drug interactions == | |||
[[Tetracycline]] and its derivatives [[demeclocycline]], [[doxycycline]], [[methacycline]], [[minocycline]], [[oxytetracycline]], [[rolitetracycline]], and [[tetracycline]] are antagonists of penicillins. The effects of oral contraceptives, including [[ethinyl estradiol] and [[mestranol]] are decreased when using penicillin. Penicillins increases the effect and toxicity of [[methotrexate]].[http://www.drugbank.ca/cgi-bin/getCard.cgi?CARD=DB00417.txt] | |||
== External links == | == External links == | ||
* {{DailyMed}} | * {{DailyMed}} | ||
* Drug Bank [http://www.drugbank.ca/cgi-bin/getCard.cgi?CARD=DB00417.txt] | * Drug Bank [http://www.drugbank.ca/cgi-bin/getCard.cgi?CARD=DB00417.txt] | ||
Revision as of 15:31, 11 February 2008
Penicillin V is a broad-spectrum, beta-lactam-based antibiotic used to treat mild to severe infections due to gram-positive bacteria. Is used to treat dental, hear, middle ear, respiratory tract and skin infections, and can also treat rheumatic and scarlet fevers.
Mechanism of action
Like other penicillin-like drugs, penicillin V works by binding to specific penicillin-binding proteins in bacterial cell walls and blocking the final cross-linking step in the synthesis of bacterial cell walls. This induces autolysis of the bactertial cells by autolysins.
Chemistry
Penicillin V is stable against degradation by beta-lactamases, including penicillinases, and cephalosporinases. Its IUPAC chemical name is (2S,5R,6R)-3,3-dimethyl-7-oxo-6-[[2-(phenoxy)acetyl]amino]-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylic acid and it has molecular formula C16H18N2O5S (MW = 350.3895 g/mol).
Drug interactions
Tetracycline and its derivatives demeclocycline, doxycycline, methacycline, minocycline, oxytetracycline, rolitetracycline, and tetracycline are antagonists of penicillins. The effects of oral contraceptives, including [[ethinyl estradiol] and mestranol are decreased when using penicillin. Penicillins increases the effect and toxicity of methotrexate.[1]
External links
- Penicillin V - FDA approved drug information (drug label) from DailyMed (U.S. National Library of Medicine).
- Drug Bank [2]