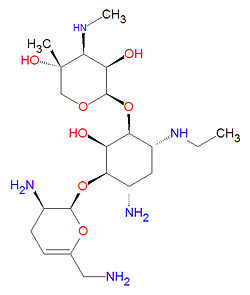
Netilmicin
Netilmicin, also called 1-N-Ethylsisomicin, is an aminoglycoside antibiotic derivative of sisomycin, an aminoglycoside antibiotic with action similar to gentamicin, but with less ear and kidney toxicity. Like other aminoglycosides, it is useful for the treatment of Gram-negative aerobic bacteria including acinetobacter, enterobacter and pseudomonas. It is active against a wide range of pathogens including E. coli, the Klebsiella-Enterobacter-Serratia bacterial group, Citrobacter sp., Proteus sp. including Proteus mirabilis, P. morganii, P. rettgrei, P. vulgaris, Pseudomonas aeruginosa and Neisseria gonorrhoea. It can be used against some bacteria that are resistant to other aminoglycosides, such as gentamicin, kanamycin, sisomicin and tobramycin, including methicillin-resistant strains of Staphylococcus (MSRA). Netilmicin is also active in vitro against isolates of Hemophilus influenzae, Salmonella sp. and Shigella sp. Some strains of Providencia sp., Acinetobacter sp. and Aeromonas sp. are also sensitive to netilmicin.
Drug interactions
The risk of kidney toxicity is increased when netilmicin is taken with cefamandole cefazolin, cefonicid, cefoperazone, ceforanide, cefotaxime, cefotetan, cefoxitin, ceftazidime, ceftizoxime, ceftriaxone, cefuroxime, cephalothin, cephapirin, cisplatin and cefradine. The effects of many muscle relaxants, including atracurium, doxacurium, gallamine triethiodide, metocurine, mivacurium, pancuronium, pipecuronium, rocuronium, succinylcholine, tubocurarine and vecuronium, are increased when taken with netilmicin. Increased ototoxicity is noted when netilmicin is taken with bumetanide, ethacrynic acid, furosemide and torasemide. Thalidomide increases the renal toxicity of metilmicin.
Mechanism of action
Aminoglycosides bind to specific 30S-subunit proteins and 16S rRNA. Specifically, netilmicin binds to four nucleotides of 16S rRNA and one amino acid of protein S12, interfering with decoding around nucleotide 1400 of 16S thereby inhibiting the formation of an initiation complex, causing misreading of mRNA and the insertion of incorrect amino acids into the polypeptide.
Its IUPAC chemical name is (2R,3R,4R,5R)-2-[(1S,2S,3R,4S,6R)-4-amino-3-[[(2S,3R)-3-amino-6-(aminomethyl) -3,4-dihydro-2H-pyran-2-yl]oxy]-6-ethylamino-2-hydroxycyclohexyl]oxy-5-methyl-4-methylaminooxane-3,5-diol and it has chemical formula C21H41N5O7. It is produced by the fermentation of Micromonospora inyoensis, a species of actinomycete, and is sold under the brand name Netromycin®.
External links
The most up-to-date information about Netilmicin and other drugs can be found at the following sites.
- Netilmicin - FDA approved drug information (drug label) from DailyMed (U.S. National Library of Medicine).
- Netilmicin - Drug information for consumers from MedlinePlus (U.S. National Library of Medicine).
- Netilmicin - Detailed information from DrugBank.