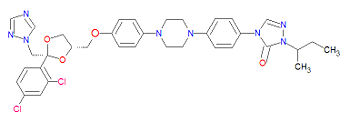
Itraconazole
|
| |||||||
| itraconazole | |||||||
| |||||||
| Uses: | antifungal drug | ||||||
| Properties: | triazole compound | ||||||
| Hazards: | see drug interactions | ||||||
| |||||||
Itraconazole is an azole-based antifungal drug containing three azoles, like fluconazole. It inhibits cytochrom P450-dependent enzymes and impairs the biosynthesis of ergosterol. It can be used to treate apergillosis, blasoplasmosis, cryptococcal meningitis, histoplasmosis and onychomycosis.
Mechanism of action
Like other azole-based antifungal agents, itraconazole interacts with 14--demethylase, a cytochrome P-450 enzyme necessary to convert lanosterol to ergosterol, an essential component of fungal cell membranes, by stopping sterol C-14 -demethylation. Itraconazole may also inhibit endogenous respiration, interact with membrane phospholipids, inhibit the transformation of yeasts to mycelial forms, inhibit purine uptake, and impair triglyceride and/or phospholipid biosynthesis[1].
Drug interactions
The azole-based antifungal agents have many drug interactions and some can be sever. This section is under construction, see ketoconazole for a partial list of drug interactions.
Chemical information
The IUPAC name for itraconazole is 2-butan-2-yl-4-[4-[4-[4-[[(2R,4S)-2-(2,4-dichlorophenyl)-2-(1,2,4-triazol-1-ylmethyl)-1,3-dioxolan-4-yl]methoxy]phenyl]piperazin-1-yl]phenyl]-1,2,4-triazol-3-one, and its chemical formula, C35H38Cl2N8O4, gives a molecular mass of 705.6334 g/mol.
Synonyms
- ITC
- ITCZ
- ITR
- Itraconazol
- Itraconazolum
- ITZ
Brand names
- Itrizole®
- Oriconazole®
- Sporal®
- Sporanos®
- Sporanox®
- Sporonox®
- Triasporn®
References and external links
- ↑ The most up-to-date information about Itraconzole and other drugs can be found at the following sites.
- Itraconzole - FDA approved drug information (drug label) from DailyMed (U.S. National Library of Medicine).
- Itraconzole - Drug information for consumers from MedlinePlus (U.S. National Library of Medicine).
- Itraconzole - Detailed information from DrugBank.