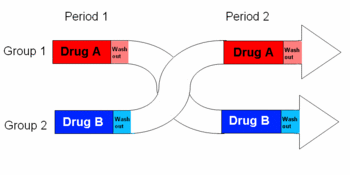
Cross-over study
In epidemiology, cross-over studies or crossover trials are a type of randomized controlled trial. Cross-over studies are "studies comparing two or more treatments or interventions in which the subjects or patients, upon completion of the course of one treatment, are switched to another. In the case of two treatments, A and B, half the subjects are randomly allocated to receive these in the order A, B and half to receive them in the order B, A. A criticism of this design is that effects of the first treatment may carry over into the period when the second is given."[1][2][3]
Special issues in the reporting of cross-over trials are:[2]
- Sequence generation
- Washout period
- Testing for carry-over effect
- Testing for period effect
- Testing for treatment effect
- Adjustment for period effect and maybe for carry-over effect
- Paired analysis
- Patient preference regarding intervention
"If carry over is detected convention suggests this may be dealt with in the analysis in one of two ways. The usual approach is to treat the study as though it were a parallel group trial and confine analysis to the first period alone."[3]
The results of crossover studies "tend to agree with those of parallel arm trials, although there was a trend for more conservative treatment effect estimates in parallel arm trials".[4]
Variations on the standard AB, BA design have been proposed.[5][6][7][8]
References
- ↑ Anonymous (2023), Cross-over studies (English). Medical Subject Headings. U.S. National Library of Medicine.
- ↑ 2.0 2.1 Mills EJ, Chan AW, Wu P, Vail A, Guyatt GH, Altman DG (2009). "Design, analysis, and presentation of crossover trials.". Trials 10: 27. DOI:10.1186/1745-6215-10-27. PMID 19405975. PMC PMC2683810. Research Blogging.
- ↑ 3.0 3.1 Sibbald B, Roberts C (1998). "Understanding controlled trials. Crossover trials.". BMJ 316 (7146): 1719. PMID 9614025. PMC PMC1113275.
- ↑ Lathyris DN, Trikalinos TA, Ioannidis JP (2007). "Evidence from crossover trials: empirical evaluation and comparison against parallel arm trials.". Int J Epidemiol 36 (2): 422-30. DOI:10.1093/ije/dym001. PMID 17301102. Research Blogging.
- ↑ Laska E, Meisner M, Kushner HB (December 1983). "Optimal crossover designs in the presence of carryover effects". Biometrics 39 (4): 1087–91. PMID 6671121. [e]
- ↑ Boon PC, Roes KC (March 1999). "Design and analysis issues for crossover designs in phase I clinical studies". J Biopharm Stat 9 (1): 109–28. PMID 10091913. [e]
- ↑ Ebbutt AF (March 1984). "Three-period crossover designs for two treatments". Biometrics 40 (1): 219–24. PMID 6733230. [e]
- ↑ Matthews JN (December 1994). "Multi-period crossover trials". Stat Methods Med Res 3 (4): 383–405. PMID 7889228. [e]